Therapeutic activation of endothelial sphingosine-1-phosphate receptor 1 by chaperone-bound S1P suppresses proliferative retinal neovascularization
- PMID: 36912000
- PMCID: PMC10165359
- DOI: 10.15252/emmm.202216645
Therapeutic activation of endothelial sphingosine-1-phosphate receptor 1 by chaperone-bound S1P suppresses proliferative retinal neovascularization
Abstract
Sphingosine-1-phosphate (S1P), the circulating HDL-bound lipid mediator that acts via S1P receptors (S1PR), is required for normal vascular development. The role of this signaling axis in vascular retinopathies is unclear. Here, we show in a mouse model of oxygen-induced retinopathy (OIR) that endothelial overexpression of S1pr1 suppresses while endothelial knockout of S1pr1 worsens neovascular tuft formation. Furthermore, neovascular tufts are increased in Apom-/- mice which lack HDL-bound S1P while they are suppressed in ApomTG mice which have more circulating HDL-S1P. These results suggest that circulating HDL-S1P activation of endothelial S1PR1 suppresses neovascular pathology in OIR. Additionally, systemic administration of ApoM-Fc-bound S1P or a small-molecule Gi-biased S1PR1 agonist suppressed neovascular tuft formation. Circulating HDL-S1P activation of endothelial S1PR1 may be a key protective mechanism to guard against neovascular retinopathies that occur not only in premature infants but also in diabetic patients and aging people.
Keywords: Sphingosine-1-phosphate (S1P); neovascularization; vascular retinopathy.
© 2023 The Authors. Published under the terms of the CC BY 4.0 license.
Conflict of interest statement
Three authors (TH, LEHS, and SS) are inventors in the patent applications for ApoM‐Fc‐S1P (WO2018052615A1).
Figures

Schematic representation of the breeding strategy to establish S1pr1 ECTG: females carrying the (ROSA)26Sortm1(CAG‐S1pr1) transgene were crossed to Cdh5‐Cre ERT2 Cre males.
Strategy to induce expression of S1PR1 in post‐natal endothelium. Pups were given tamoxifen at P12 and P14, and retinas were analyzed at P17.
Flat‐mounted retinas from OIR S1pr1 f/stop/f and S1pr1 ECTG pups at P17. High‐magnification pictures of superficial capillaries showing S1PR1 induction in the S1pr1 ECTG.
Flat‐mounted retinas from OIR S1pr1 f/stop/f and S1pr1 ECTG pups at P17. Retinal vasculature parameters were stained with isolectin and images of superficial (top panel), intermediary (middle panel), and deep (lower panel) plexuses are shown.
Quantification of morphometric parameters in between retinas from S1pr1 f/stop/f and S1pr1 ECTG. Retinas from P9 S1pr1f/stop/f mice were used as controls.
Dot plot representing expression level and frequency of cell types markers among non‐neuronal retinal cells at P14 and P17 in OIR.
Volcano plot showing expression level and frequency of OIR‐induced genes (left) and S1P‐related genes (right) among non‐neuronal retinal cells at P14 (left) and P17 (right) in OIR. S1pr4 and S1pr5 expression was limited to a low number of endothelial cells.
Cross sections from OIR S1pr1 f/stop/f , S1pr1 ECTG, and S1pr1 ECKO P17 pups stained for S1PR1 (red). Blood vessels are delineated by collagen IV (green) and nuclei are stained with Hoechst (blue) (left). Quantification of associated S1PR1 fluorescence inside blood vessels (right).
VEGF level in P17 retinas quantified by ELISA. Retinas from normoxic S1pr1 f/stop/f animal (n = 10) or OIR S1pr1 f/stop/f (n = 10) and S1pr1 ECTG pups (n = 13) at P17 were used.
Flat‐mounted retinas from OIR S1pr1 f/stop/f and S1pr1 ECTG pups at P15. Blood vessels are stained with isolectin (left). Avascular area, total neovascular tuft area, and average neovascular tuft size are quantified (right).
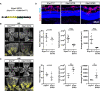
Strategy to induce S1PR1 expression in the endothelium post‐OIR. S1pr1 ECTG pups were exposed to 75% oxygen from P7 to P12. Upon return to normoxia, S1PR1 expression was induced by tamoxifen injection at P12 and P14, and retinas were analyzed at P17.
Flat‐mounted retinas from OIR S1pr1 f/stop/f and S1pr1 ECTG pups at P17. Blood vessels are stained with isolectin (left) and pathological neovascular tuft area is highlighted in yellow. Avascular area and total and average neovascular tuft areas are quantified (right)
Flat‐mounted retinas from OIR S1pr1 f/f and S1pr1 ECKO pups, at P17. Blood vessels are stained with isolectin and neovascular tufts are highlighted in yellow. Avascular area and total and average neovascular tuft areas are quantified.
Cross sections from OIR S1pr1 f/stop/f , S1pr1 ECTG, and S1pr1 ECKO P17 pups. Blood vessels are stained with isolectin (red) and nuclei with Hoechst (blue).

Retinal flat mounts from OIR S1pr1 f/stop/f or S1pr1 ECTG or ECKO P17 pups. High‐magnification view of neovascular tufts stained for VE‐cadherin (green) and Claudin‐5 (red). Junctional levels of VE‐cadherin and Claudin‐5 were quantified as described, for a minimum of five animals per genotype.
Retinal flat mounts from OIR S1pr1 f/stop/f or S1pr1 ECTG or ECKO P17 pups. Sites of vascular leakage were assessed by staining for fibrinogen (red), and blood vessels are delineated by isolectin (green). Extravascular fibrinogen was quantified as described for a minimum of five animals per genotype.
Pericytes stained by NG2 on the surface of isolectin‐positive neovascular tufts at P17 post‐OIR (left). EC‐associated pericytes in neovascular tufts were quantified (right). A total of 70 tufts from three different animals were assessed.
Flat‐mounted retinas from OIR S1pr1 f/f and S1pr1 ECKO pups at P21 stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified. A minimum of seven pups were analyzed per group.

Flat‐mount view of neovascular tufts from S1pr1 f/stop/f , S1pr1 ECTG, or ECKO at P17, stained for endothelial nuclei (ERG), blood vessel (isolectin), and associated endothelial nuclei quantification.
Flat‐mount view of neovascular tufts from S1pr1 f/stop/f or S1pr1 ECTG at P17, stained for immune cells (CD45), blood vessels (isolectin) (left), and FACS on retinal single‐cell preparations (right). Quantification of immune cells by FACS in OIR retinas from S1pr1 f/stop/f and S1pr1 ECTG at P17. Total immune cells (CD45 positive, left panel), monocytes/macrophages (CD11b positive, middle panel), and neutrophils (CD11b and Ly6G double positive, right panel) are presented.
Flat‐mounted retinas from OIR S1pr1 f/stop/f and S1pr1 ECTG pups at P19 stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified.
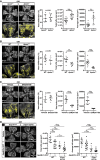
Flat‐mounted retinas from OIR Apom +/− and Apom −/− pups at P17, stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified (n = 10 pups/group).
Flat‐mounted retinas from OIR WT and Apom TG pups at P17, stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified (n = 7 pups/group).
Flat‐mounted retinas from SAR247799‐treated, OIR WT pups at P17, stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified (n = 10 pups/group).
Flat‐mounted retinas from ApoM‐Fc‐S1P‐treated, OIR S1pr1 f/stop/f , or S1pr1 ECTG pups at P17, stained with isolectin. Avascular area and total and average neovascular tuft areas are quantified (n = 6 pups/group).

VEGF expression level in P17 retinas quantified by ELISA. Retinas from post‐OIR vehicle‐ or ApoM‐Fc‐S1P‐treated (n = 7 and 8, respectively) WT pups at P17.
Retinal flat mounts from OIR vehicle‐ or ApoM‐Fc‐S1P‐treated pups. High‐magnification view of neovascular tufts stained for VE‐cadherin (green) and Claudin‐5 (red). Junctional density was quantified as described by a minimum of seven animals per condition.
References
-
- Bakri SJ, Moshfeghi DM, Francom S, Rundle AC, Reshef DS, Lee PP, Schaeffer C, Rubio RG, Lai P (2014) Intraocular pressure in eyes receiving monthly ranibizumab in 2 pivotal age‐related macular degeneration clinical trials. Ophthalmology 121: 1102–1108 - PubMed
-
- Ben Shoham A, Malkinson G, Krief S, Shwartz Y, Ely Y, Ferrara N, Yaniv K, Zelzer E (2012) S1P1 inhibits sprouting angiogenesis during vascular development. Development 139: 3859–3869 - PubMed
-
- Bergougnan L, Armani S, Golor G, Tardat A, Vitse O, Hurbin F, Scemama M, Poitiers F, Radzik D, Gaudin C et al (2021) First‐in‐human study of the safety, tolerability, pharmacokinetics and pharmacodynamics of single and multiple oral doses of SAR247799, a selective G‐protein‐biased sphingosine‐1 phosphate receptor‐1 agonist for endothelial protection. Br J Clin Pharmacol 87: 598–611 - PMC - PubMed
-
- Binet F, Cagnone G, Crespo‐Garcia S, Hata M, Neault M, Dejda A, Wilson AM, Buscarlet M, Mawambo GT, Howard JP et al (2020) Neutrophil extracellular traps target senescent vasculature for tissue remodeling in retinopathy. Science 369: eaay5356 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

