Association of glucagon-like peptide-1 receptor agonist treatment with gastric residue in an esophagogastroduodenoscopy
- PMID: 36919944
- PMCID: PMC10204182
- DOI: 10.1111/jdi.14005
Association of glucagon-like peptide-1 receptor agonist treatment with gastric residue in an esophagogastroduodenoscopy
Abstract
Aims/introduction: Previous studies have reported that the glucagon-like peptide-1 receptor agonist (GLP-1RA) delays gastric emptying, and gastric emptying was assessed by the 13 C breath test or paracetamol absorption technique. However, neither of them is clinically familiar in real-world clinical practice. The purpose of the present study was to investigate the association between GLP-1RA treatment and gastric residue in an esophagogastroduodenoscopy.
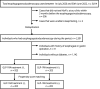
Materials and methods: This study was a matched pair case-control study. The study population consisted of 1,128 individuals with diabetes who had esophagogastroduodenoscopy at our clinic between July 2020 and June 2022. To account for differences in characteristics, such as age, sex, insulin treatment and glycated hemoglobin, we carried out a one-to-one nearest neighbor propensity score matching analysis between diabetes patients with and without GLP-1RA treatment. After matching, we compared the presence of gastric residue in an esophagogastroduodenoscopy by the McNemar test between patients with and without GLP-1RA treatment.
Results: After the propensity score matching, we selected 205 pairs. In the propensity score-matched comparison, the proportion of gastric residue was statistically significantly higher in the GLP-1RA treatment group (0.49% vs 5.4%, P = 0.004). The details of GLP-1RA prescribed for the 11 patients with gastric residue were liraglutide once daily 1.8 mg (n = 2), dulaglutide once weekly 0.75 mg (n = 5), semaglutide once weekly 0.5 mg (n = 2) and semaglutide once weekly 1.0 mg (n = 2).
Conclusion: GLP-1RA treatment is associated with gastric residue in an esophagogastroduodenoscopy in patients with diabetes.
Keywords: Esophagogastroduodenoscopy; Gastric residue; Glucagon-like peptide-1 receptor agonist treatment.
© 2023 The Authors. Journal of Diabetes Investigation published by Asian Association for the Study of Diabetes (AASD) and John Wiley & Sons Australia, Ltd.
Figures
Similar articles
-
Review of head-to-head comparisons of glucagon-like peptide-1 receptor agonists.Diabetes Obes Metab. 2016 Apr;18(4):317-32. doi: 10.1111/dom.12596. Epub 2015 Dec 29. Diabetes Obes Metab. 2016. PMID: 26511102 Free PMC article. Review.
-
Glucagon-Like Peptide-1 Receptor Agonists Increase Solid Gastric Residue Rates on Upper Endoscopy Especially in Patients With Complicated Diabetes: A Case-Control Study.Am J Gastroenterol. 2024 Jun 1;119(6):1081-1088. doi: 10.14309/ajg.0000000000002777. Epub 2024 Mar 27. Am J Gastroenterol. 2024. PMID: 38534127
-
Oral semaglutide versus injectable glucagon-like peptide-1 receptor agonists: a cost of control analysis.J Med Econ. 2020 Jun;23(6):650-658. doi: 10.1080/13696998.2020.1722678. Epub 2020 Feb 7. J Med Econ. 2020. PMID: 31990244
-
Impact on HbA1c and body weight of switching from other GLP-1 receptor agonists to semaglutide: A model-based approach.Diabetes Obes Metab. 2019 Jan;21(1):43-51. doi: 10.1111/dom.13479. Epub 2018 Aug 23. Diabetes Obes Metab. 2019. PMID: 30047216 Free PMC article. Clinical Trial.
-
GLP-1 Agonist to Treat Obesity and Prevent Cardiovascular Disease: What Have We Achieved so Far?Curr Atheroscler Rep. 2022 Nov;24(11):867-884. doi: 10.1007/s11883-022-01062-2. Epub 2022 Aug 31. Curr Atheroscler Rep. 2022. PMID: 36044100 Review.
Cited by
-
A narrative review of glucagon-like peptide-1 receptor agonists prior to deep sedation or general anesthesia.J Anesth Analg Crit Care. 2025 Mar 28;5(1):16. doi: 10.1186/s44158-025-00237-y. J Anesth Analg Crit Care. 2025. PMID: 40156058 Free PMC article. Review.
-
Glucagon-Like Peptide-1 Receptor Agonists Do Not Increase Aspiration During Upper Endoscopy in Patients With Diabetes.Clin Gastroenterol Hepatol. 2025 Apr;23(5):739-747. doi: 10.1016/j.cgh.2024.04.038. Epub 2024 May 15. Clin Gastroenterol Hepatol. 2025. PMID: 38759826
-
Real-World Impact of GLP-1 Receptor Agonists on Endoscopic Patient Outcomes in an Ambulatory Setting: A Retrospective Study at a Large Tertiary Center.J Clin Med. 2024 Sep 12;13(18):5403. doi: 10.3390/jcm13185403. J Clin Med. 2024. PMID: 39336890 Free PMC article.
-
Anesthesia and glucagon-like peptide-1 receptor agonists: proceed with caution!Can J Anaesth. 2023 Aug;70(8):1281-1286. doi: 10.1007/s12630-023-02550-y. Epub 2023 Jul 19. Can J Anaesth. 2023. PMID: 37466910 English. No abstract available.
-
Glucose-Lowering Agents Developed in the Last Two Decades and Their Perioperative Implications.Pharmaceuticals (Basel). 2024 Dec 24;18(1):4. doi: 10.3390/ph18010004. Pharmaceuticals (Basel). 2024. PMID: 39861067 Free PMC article. Review.
References
-
- Draznin B, Aroda VR, Bakris G, et al. 9. Pharmacologic approaches to glycemic treatment: Standards of medical Care in Diabetes‐2022. Diabetes Care 2022; 45: S125–s143. - PubMed
-
- Bouchi R, Kondo T, Ohta Y, et al. A proposed algorithm for pharmacotherapy in people with type 2 diabetes. J Japan Diabetes Soc 2022; 65: 419–434 (Japanese).
-
- Quast DR, Nauck MA, Schenker N, et al. Macronutrient intake, appetite, food preferences and exocrine pancreas function after treatment with short‐ and long‐acting glucagon‐like peptide‐1 receptor agonists in type 2 diabetes. Diabetes Obes Metab 2021; 23: 2344–2353. - PubMed
-
- Quast DR, Schenker N, Menge BA, et al. Effects of Lixisenatide versus Liraglutide (short‐ and long‐acting GLP‐1 receptor agonists) on esophageal and gastric function in patients with type 2 diabetes. Diabetes Care 2020; 43: 2137–2145. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical