Comparison of Toxicities among Different Bumped Kinase Inhibitor Analogs for Treatment of Cryptosporidiosis
- PMID: 36920244
- PMCID: PMC10112232
- DOI: 10.1128/aac.01425-22
Comparison of Toxicities among Different Bumped Kinase Inhibitor Analogs for Treatment of Cryptosporidiosis
Abstract
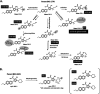
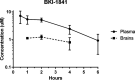
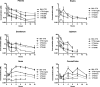
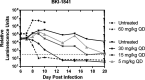
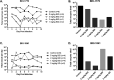
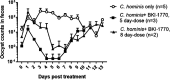
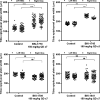
Recent advances on the development of bumped kinase inhibitors for treatment of cryptosporidiosis have focused on the 5-aminopyrazole-4-carboxamide scaffold, due to analogs that have less hERG inhibition, superior efficacy, and strong in vitro safety profiles. Three compounds, BKI-1770, -1841, and -1708, showed strong efficacy in C. parvum infected mice. Both BKI-1770 and BKI-1841 had efficacy in the C. parvum newborn calf model, reducing diarrhea and oocyst excretion. However, both compounds caused hyperflexion of the limbs seen as dropped pasterns. Toxicity experiments in rats and calves dosed with BKI-1770 showed enlargement of the epiphyseal growth plate at doses only slightly higher than the efficacious dose. Mice were used as a screen to check for bone toxicity, by changes to the tibia epiphyseal growth plate, or neurological causes, by use of a locomotor activity box. These results showed neurological effects from both BKI-1770 and BKI-1841 and bone toxicity in mice from BKI-1770, indicating one or both effects may be contributing to toxicity. However, BKI-1708 remains a viable treatment candidate for further evaluation as it showed no signs of bone toxicity or neurological effects in mice.
Keywords: BKI-1708; BKI-1770; BKI-1841; Cryptosporidium; bumped kinase inhibitors; calcium-dependent protein kinases; cryptosporidiosis; epiphyseal growth plate.
Conflict of interest statement
The authors declare a conflict of interest. Wesley C. Van Voorhis is a co-owner of ParaTheraTech Inc., a company that is developing bumped-kinase inhibitors for animal therapy. Otherwise the authors have nothing to report.
Figures
References
-
- Kotloff KL, Nataro JP, Blackwelder WC, Nasrin D, Farag TH, Panchalingam S, Wu Y, Sow SO, Sur D, Breiman RF, Faruque AS, Zaidi AK, Saha D, Alonso PL, Tamboura B, Sanogo D, Onwuchekwa U, Manna B, Ramamurthy T, Kanungo S, Ochieng JB, Omore R, Oundo JO, Hossain A, Das SK, Ahmed S, Qureshi S, Quadri F, Adegbola RA, Antonio M, Hossain MJ, Akinsola A, Mandomando I, Nhampossa T, Acacio S, Biswas K, O'Reilly CE, Mintz ED, Berkeley LY, Muhsen K, Sommerfelt H, Robins-Browne RM, Levine MM. 2013. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): a prospective, case-control study. Lancet 382:209–222. doi:10.1016/S0140-6736(13)60844-2. - DOI - PubMed
-
- Khalil IA, Troeger C, Rao PC, Blacker BF, Brown A, Brewer TG, Colombara DV, De Hostos EL, Engmann C, Guerrant RL, Haque R, Houpt ER, Kang G, Korpe PS, Kotloff KL, Lima AAM, Petri WA, Jr, Platts-Mills JA, Shoultz DA, Forouzanfar MH, Hay SI, Reiner RC, Jr, Mokdad AH. 2018. Morbidity, mortality, and long-term consequences associated with diarrhoea from Cryptosporidium infection in children younger than 5 years: a meta-analyses study. Lancet Glob Health 6:e758–e768. doi:10.1016/S2214-109X(18)30283-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

