Lipocalin-2 promotes acute lung inflammation and oxidative stress by enhancing macrophage iron accumulation
- PMID: 36923935
- PMCID: PMC10008694
- DOI: 10.7150/ijbs.79915
Lipocalin-2 promotes acute lung inflammation and oxidative stress by enhancing macrophage iron accumulation
Abstract
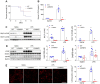
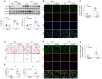
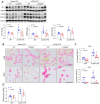
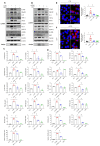
Lipocalin-2 (LCN2) is an acute-phase protein that regulates inflammatory responses to bacteria or lipopolysaccharide (LPS). Although the bacteriostatic role of LCN2 is well studied, the function of LCN2 in acute lung damage remains unclear. Here, LCN2 knockout (KO) mice were used to investigate the role of LCN2 in LPS-treated mice with or without recombinant LCN2 (rLCN2). In addition, we employed patients with pneumonia. RAW264.7 cells were given LCN2 inhibition or rLCN2 with or without iron chelator deferiprone. LCN2 KO mice had a higher survival rate than wild-type (WT) mice after LPS treatment. In addition to elevated LCN2 levels in serum and bronchoalveolar lavage fluid (BALF), LPS treatment also increased LCN2 protein in alveolar macrophage lysates of BALF. LCN2 deletion attenuated neutrophil and macrophage infiltration in the lungs of LPS-treated mice as well as serum and BALF interleukin-6 (IL-6). Circulating proinflammatory cytokines and LCN2-positive macrophages were prominently increased in the BALF of pneumonia patients. In addition to increase of iron-stained macrophages in pneumonia patients, increased iron-stained macrophages and oxidative stress in LPS-treated mice were inhibited by LCN2 deletion. In contrast, rLCN2 pretreatment aggravated lung inflammation and oxidative stress in LPS-treated WT mice and then resulted in higher mortality. In RAW264.7 cells, exogenous LCN2 treatment also increased inflammation and oxidative stress, whereas LCN2 knockdown markedly diminished these effects. Furthermore, deferiprone inhibited inflammation, oxidative stress, and phagocytosis in RAW264.7 cells with high LCN2 levels, as well as LPS-induced acute lung injury in WT and LCN2 KO mice. Thus, these findings suggest that LCN2 plays a key role in inflammation and oxidative stress following acute lung injury and that LCN2 is a potential therapeutic target for pneumonia or acute lung injury.
Keywords: Acute lung injury; Inflammation; Iron; Lipocalin-2; Macrophage; Oxidative stress.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures


References
-
- Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A. et al. Epidemiology, Patterns of Care, and Mortality for Patients With Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries. Jama. 2016;315:788–800. - PubMed
-
- Cilloniz C, Ferrer M, Liapikou A, Garcia-Vidal C, Gabarrus A, Ceccato A, Acute respiratory distress syndrome in mechanically ventilated patients with community-acquired pneumonia. Eur Respir J. 2018. 51. - PubMed
-
- Goerdt S, Politz O, Schledzewski K, Birk R, Gratchev A, Guillot P. et al. Alternative versus classical activation of macrophages. Pathobiology. 1999;67:222–6. - PubMed
-
- Gordon S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3:23–35. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

