Artificial intelligence-based analysis of whole-body bone scintigraphy: The quest for the optimal deep learning algorithm and comparison with human observer performance
- PMID: 36932023
- PMCID: PMC11156776
- DOI: 10.1016/j.zemedi.2023.01.008
Artificial intelligence-based analysis of whole-body bone scintigraphy: The quest for the optimal deep learning algorithm and comparison with human observer performance
Abstract
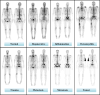
Purpose: Whole-body bone scintigraphy (WBS) is one of the most widely used modalities in diagnosing malignant bone diseases during the early stages. However, the procedure is time-consuming and requires vigour and experience. Moreover, interpretation of WBS scans in the early stages of the disorders might be challenging because the patterns often reflect normal appearance that is prone to subjective interpretation. To simplify the gruelling, subjective, and prone-to-error task of interpreting WBS scans, we developed deep learning (DL) models to automate two major analyses, namely (i) classification of scans into normal and abnormal and (ii) discrimination between malignant and non-neoplastic bone diseases, and compared their performance with human observers.
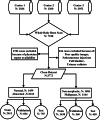
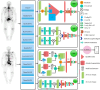
Materials and methods: After applying our exclusion criteria on 7188 patients from three different centers, 3772 and 2248 patients were enrolled for the first and second analyses, respectively. Data were split into two parts, including training and testing, while a fraction of training data were considered for validation. Ten different CNN models were applied to single- and dual-view input (posterior and anterior views) modes to find the optimal model for each analysis. In addition, three different methods, including squeeze-and-excitation (SE), spatial pyramid pooling (SPP), and attention-augmented (AA), were used to aggregate the features for dual-view input models. Model performance was reported through area under the receiver operating characteristic (ROC) curve (AUC), accuracy, sensitivity, and specificity and was compared with the DeLong test applied to ROC curves. The test dataset was evaluated by three nuclear medicine physicians (NMPs) with different levels of experience to compare the performance of AI and human observers.
Results: DenseNet121_AA (DensNet121, with dual-view input aggregated by AA) and InceptionResNetV2_SPP achieved the highest performance (AUC = 0.72) for the first and second analyses, respectively. Moreover, on average, in the first analysis, Inception V3 and InceptionResNetV2 CNN models and dual-view input with AA aggregating method had superior performance. In addition, in the second analysis, DenseNet121 and InceptionResNetV2 as CNN methods and dual-view input with AA aggregating method achieved the best results. Conversely, the performance of AI models was significantly higher than human observers for the first analysis, whereas their performance was comparable in the second analysis, although the AI model assessed the scans in a drastically lower time.
Conclusion: Using the models designed in this study, a positive step can be taken toward improving and optimizing WBS interpretation. By training DL models with larger and more diverse cohorts, AI could potentially be used to assist physicians in the assessment of WBS images.
Keywords: Artificial intelligence; Bone; Deep learning; Scintigraphy; Whole-body.
Copyright © 2023 The Author(s). Published by Elsevier GmbH.. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






Similar articles
-
Deep learning model using planar whole-body bone scintigraphy for diagnosis of skull base invasion in patients with nasopharyngeal carcinoma.J Cancer Res Clin Oncol. 2024 Oct 9;150(10):449. doi: 10.1007/s00432-024-05969-y. J Cancer Res Clin Oncol. 2024. PMID: 39379746 Free PMC article.
-
Deep neural network based artificial intelligence assisted diagnosis of bone scintigraphy for cancer bone metastasis.Sci Rep. 2020 Oct 12;10(1):17046. doi: 10.1038/s41598-020-74135-4. Sci Rep. 2020. PMID: 33046779 Free PMC article.
-
Generative artificial intelligence enables the generation of bone scintigraphy images and improves generalization of deep learning models in data-constrained environments.Eur J Nucl Med Mol Imaging. 2025 Jun;52(7):2355-2368. doi: 10.1007/s00259-025-07091-8. Epub 2025 Jan 29. Eur J Nucl Med Mol Imaging. 2025. PMID: 39878897 Free PMC article.
-
Automatic identification of suspicious bone metastatic lesions in bone scintigraphy using convolutional neural network.BMC Med Imaging. 2021 Sep 4;21(1):131. doi: 10.1186/s12880-021-00662-9. BMC Med Imaging. 2021. PMID: 34481459 Free PMC article.
-
Recent advances in antibody optimization based on deep learning methods.J Zhejiang Univ Sci B. 2025 May 28;26(5):409-420. doi: 10.1631/jzus.B2400387. J Zhejiang Univ Sci B. 2025. PMID: 40436639 Free PMC article. Review.
Cited by
-
Application of Nuclear Medicine Techniques in Musculoskeletal Infection: Current Trends and Future Prospects.J Clin Med. 2024 Feb 13;13(4):1058. doi: 10.3390/jcm13041058. J Clin Med. 2024. PMID: 38398371 Free PMC article. Review.
-
Artificial intelligence in medical physics.Z Med Phys. 2024 May;34(2):177-178. doi: 10.1016/j.zemedi.2024.03.002. Epub 2024 Mar 23. Z Med Phys. 2024. PMID: 38523040 Free PMC article. No abstract available.
-
Artificial intelligence-based cardiac transthyretin amyloidosis detection and scoring in scintigraphy imaging: multi-tracer, multi-scanner, and multi-center development and evaluation study.Eur J Nucl Med Mol Imaging. 2025 Jun;52(7):2513-2528. doi: 10.1007/s00259-025-07117-1. Epub 2025 Feb 5. Eur J Nucl Med Mol Imaging. 2025. PMID: 39907796 Free PMC article.
-
Does the tail show when the nose knows? Artificial intelligence outperforms human experts at predicting detection dogs finding their target through tail kinematics.R Soc Open Sci. 2025 Aug 13;12(8):250399. doi: 10.1098/rsos.250399. eCollection 2025 Aug. R Soc Open Sci. 2025. PMID: 40809360 Free PMC article.
-
Clinical performance of deep learning-enhanced ultrafast whole-body scintigraphy in patients with suspected malignancy.BMC Med Imaging. 2024 Sep 9;24(1):236. doi: 10.1186/s12880-024-01422-1. BMC Med Imaging. 2024. PMID: 39251959 Free PMC article.
References
-
- O'Connor M.K., Brown M.L., Hung J.C., Hayostek R.J. The art of bone scintigraphy–technical aspects. J Nucl Med. 1991;32:2332–2341. - PubMed
-
- Savelli G., Maffioli L., Maccauro M., De Deckere E., Bombardieri E. Bone scintigraphy and the added value of SPECT (single photon emission tomography) in detecting skeletal lesions. Q J Nucl Med. 2001;45:27–37. - PubMed
-
- Fogelman I., Gnanasegaran G. Springer; 2013. Van der Wall H. Radionuclide and hybrid bone imaging. - PubMed
-
- Ryan P., Fogelman I. Bone scintigraphy in metabolic bone disease. Semin Nucl Med. 1997:291–305. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

