Adenoviral vectored vaccination protects against Crimean-Congo Haemorrhagic Fever disease in a lethal challenge model
- PMID: 36933409
- PMCID: PMC10025009
- DOI: 10.1016/j.ebiom.2023.104523
Adenoviral vectored vaccination protects against Crimean-Congo Haemorrhagic Fever disease in a lethal challenge model
Abstract
Background: The tick-borne bunyavirus, Crimean-Congo Haemorrhagic Fever virus (CCHFV), can cause severe febrile illness in humans and has a wide geographic range that continues to expand due to tick migration. Currently, there are no licensed vaccines against CCHFV for widespread usage.
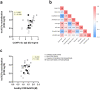
Methods: In this study, we describe the preclinical assessment of a chimpanzee adenoviral vectored vaccine (ChAdOx2 CCHF) which encodes the glycoprotein precursor (GPC) from CCHFV.
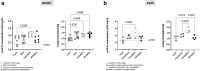
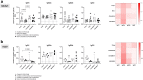
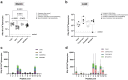
Findings: We demonstrate here that vaccination with ChAdOx2 CCHF induces both a humoral and cellular immune response in mice and 100% protection in a lethal CCHF challenge model. Delivery of the adenoviral vaccine in a heterologous vaccine regimen with a Modified Vaccinia Ankara vaccine (MVA CCHF) induces the highest levels of CCHFV-specific cell-mediated and antibody responses in mice. Histopathological examination and viral load analysis of the tissues of ChAdOx2 CCHF immunised mice reveals an absence of both microscopic changes and viral antigen associated with CCHF infection, further demonstrating protection against disease.
Interpretation: There is the continued need for an effective vaccine against CCHFV to protect humans from lethal haemorrhagic disease. Our findings support further development of the ChAd platform expressing the CCHFV GPC to seek an effective vaccine against CCHFV.
Funding: This research was supported by funding from the Biotechnology and Biological Sciences Research Council (UKRI-BBSRC) [BB/R019991/1 and BB/T008784/1].
Keywords: Antibodies; CCHFV; ChAdOx2; Heterologous immunisation; Prime and boost; T cells; Vaccine.
Copyright © 2023 The Author(s). Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of interests SCG is co-founder and board member of Vaccitech and named as an inventor on a patent covering use of ChAdOx2-vectored vaccines. TL was consultant to Vaccitech. The remaining authors declare no competing interests.
Figures




References
-
- Walter C.T., Barr J.N. Recent advances in the molecular and cellular biology of bunyaviruses. J Gen Virol. 2011;92(Pt 11):2467–2484. - PubMed
-
- Bente D.A., Forrester N.L., Watts D.M., McAuley A.J., Whitehouse C.A., Bray M. Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity. Antiviral Res. 2013;100(1):159–189. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

