Progesterone, cerclage, pessary, or acetylsalicylic acid for prevention of preterm birth in singleton and multifetal pregnancies - A systematic review and meta-analyses
- PMID: 36936217
- PMCID: PMC10015499
- DOI: 10.3389/fmed.2023.1111315
Progesterone, cerclage, pessary, or acetylsalicylic acid for prevention of preterm birth in singleton and multifetal pregnancies - A systematic review and meta-analyses
Erratum in
-
Corrigendum: Progesterone, cerclage, pessary, or acetylsalicylic acid for prevention of preterm birth in singleton and multifetal pregnancies - A systematic review and meta-analyses.Front Med (Lausanne). 2025 Feb 14;12:1561413. doi: 10.3389/fmed.2025.1561413. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40027900 Free PMC article.
Abstract
Background: Preterm birth is the leading cause of childhood mortality and morbidity. We aimed to provide a comprehensive systematic review on randomized controlled trials (RCTs) on progesterone, cerclage, pessary, and acetylsalicylic acid (ASA) to prevent preterm birth in asymptomatic women with singleton pregnancies defined as risk of preterm birth and multifetal pregnancies.
Methods: Six databases (including PubMed, Embase, Medline, the Cochrane Library) were searched up to February 2022. RCTs published in English or Scandinavian languages were included through a consensus process. Abstracts and duplicates were excluded. The trials were critically appraised by pairs of reviewers. The Cochrane risk-of-bias tool was used for risk of bias assessment. Predefined outcomes including preterm birth, perinatal/neonatal/maternal mortality and morbidity, were pooled in meta-analyses using RevMan 5.4, stratified for high and low risk of bias trials. The certainty of evidence was assessed using the GRADE approach. The systematic review followed the PRISMA guideline.
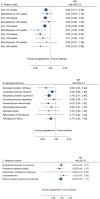
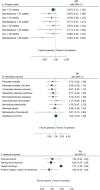
Results: The search identified 2,309 articles, of which 87 were included in the assessment: 71 original RCTs and 16 secondary publications with 23,886 women and 32,893 offspring. Conclusions were based solely on trials with low risk of bias (n = 50).Singleton pregnancies: Progesterone compared with placebo, reduced the risk of preterm birth <37 gestational weeks: 26.8% vs. 30.2% (Risk Ratio [RR] 0.82 [95% Confidence Interval [CI] 0.71 to 0.95]) (high certainty of evidence, 14 trials) thereby reducing neonatal mortality and respiratory distress syndrome. Cerclage probably reduced the risk of preterm birth <37 gestational weeks: 29.0% vs. 37.6% (RR 0.78 [95% CI 0.69 to 0.88]) (moderate certainty of evidence, four open trials). In addition, perinatal mortality may be reduced by cerclage. Pessary did not demonstrate any overall effect. ASA did not affect any outcome, but evidence was based on one underpowered study.Multifetal pregnancies: The effect of progesterone, cerclage, or pessary was minimal, if any. No study supported improved long-term outcome of the children.
Conclusion: Progesterone and probably also cerclage have a protective effect against preterm birth in asymptomatic women with a singleton pregnancy at risk of preterm birth. Further trials of ASA are needed. Prevention of preterm birth requires screening programs to identify women at risk of preterm birth.
Systematic review registration: [https://www.crd.york.ac.uk/prospero/], identifier [CRD42021234946].
Keywords: acetylsalicylic acid; cerclage; perinatal morbidity and mortality; pessary; preterm birth; progesterone; systematic review.
Copyright © 2023 Wennerholm, Bergman, Kuusela, Ljungström, Möller, Hongslo Vala, Ekelund, Liljegren, Petzold, Sjögren, Svensson, Strandell and Jacobsson.
Conflict of interest statement
LB board member and responsible for the biobank in the IMPACT study where PlGF reagents have been donated by Roche, Perkin Elmer, and Thermo Fischer. Course leader for the course in Preeclampsia in Sweden with sponsorship by Thermo Fischer and Roche. Obtained reimbursement for lecture by iLab Medical and reimbursement as expert opinion from Homburg and Partner. Board member in intervention trials where the trial drug is donated by Merck. Associate member in FIGO for the working group of maternal longterm health. Chair of the Swedish Preeclampsia Working Group. Board member in CoLab. Author of the chapter about preeclampsia in “Obstetrik” and about pulmonary hypertension in “Internetmedicin.” Research grants from the Swedish Research Council, STINT, Märta Lundqvist stiftelse, Swedish Society of Medicine, Gothenburg Society of Medicine, SSMF, Jane and Dan Olssons Stiftelse, Swedish Brain fund, Jeanssons stiftelse and Wallenberg Center for Molecular and Translational Medicine. BJ Research grants from Swedish Research Council, Norwegian Research Council, March of Dimes, Burroughs Wellcome Fund and the US National Institute of Health. During the last years performed clinical diagnostic trials on NIPT with Ariosa (completed), Natera (ongoing), Vanadis (completed), and Hologic (ongoing) with expenditures reimbursed per patient. Also performed clinical probiotic studies with the probiotic product provided by FukoPharma (ongoing, no funding) and BioGaia (ongoing; also provided a research grant for the specific study). Previously (2018–2020) a collaborator in IMPACT study where Roche, Perkin Elmer and Thermo Fisher provided reagents to PLGF analyses. No lectures, presentations, travel or personal reimbursement has been financed by any company. Division Director of Maternal and Neonatal Health of the International Federation of Gynecology and Obstetrics (FIGO), Board member of the European Association of Perinatal Medicine and chair the EAPM special interest group of preterm delivery. Steering group member of Genomic Medicine Sweden and is in charge of the Genomic Medicine Sweden complex diseases group. Swedish representative in Nordic Society of Precision Medicine. U-BW, BJ, and PK declare being co-investigators in the OPPTIMUM trial, an included trial in this review. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Comment in
-
Excerpts from the World Medical Literature: Obstetrics.J Obstet Gynaecol Can. 2023 Jul;45(7):479-481. doi: 10.1016/j.jogc.2023.04.018. J Obstet Gynaecol Can. 2023. PMID: 37400184 No abstract available.
References
-
- Jacobsson B, Pettersson K, Modzelewska D, Abrahamsson T, Bergman L, Håkansson S. Förtidsbörd största perinatala problemet - 5,7 procent av graviditeter i Sverige slutar för tidigt, inte klarlagt varför – kostar miljardbelopp varje år [Preterm delivery: an overview on epidemiology, pathophysiology and consequences for the individual and the society]. Lakartidningen. (2019) 116:FR6F, PMID: - PubMed
Publication types
LinkOut - more resources
Full Text Sources

