Protective effect of L‑carnitine against oxidative stress injury in human ovarian granulosa cells
- PMID: 36936706
- PMCID: PMC10015319
- DOI: 10.3892/etm.2023.11860
Protective effect of L‑carnitine against oxidative stress injury in human ovarian granulosa cells
Abstract
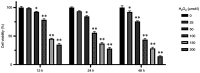
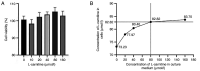
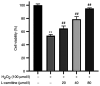
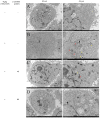
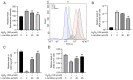
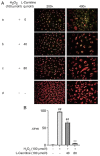
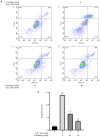
Granulosa cells (GCs) are important for supporting and nourishing oocytes during follicular development and maturation. Oxidative stress (OS) injury of GCs can lead to decreased responsiveness of follicles to follicular stimulating hormone (FSH), which will accelerate ovarian senescence and adversely affect oocyte and embryo quality. Since L-carnitine has been previously reported to exert strong antioxidant activity, the present study aimed to explore the possible effects of L-carnitine on OS injury and FSH receptor (FSHR) expression in ovarian GCs, results of which may be of significance for GCs protection. In the present study, OS was induced in vitro in KGN cells by treatment with H2O2. KGN cells were cultured and divided into the following four groups: Blank, OS, and 40 and 80 µmol/l L-carnitine pre-treatment groups. In the OS group, cells showed nuclear pyknosis, mitochondria swelled irregularly whilst featuring fractured cristae. In addition, cell viability, ROS levels, superoxide dismutase levels, glutathione levels, malondialdehyde levels, the mitochondrial membrane potential and FSHR expression, as determined by Cell Counting Kit-8 (CCK-8), 2,7-dichloro-dihydrofluorescein diacetate, spectrophotometry, ELISA, spectrophotometry, JC-1 and western blot analyses, respectively, were all significantly different in the OS group compared with those in the control group. However, malonaldehyde levels, reactive oxygen species levels and the apoptosis rate according to flow cytometry were all significantly increased compared with those in the control. Compared with those in the OS group, the morphology of cells and mitochondria in the L-carnitine pre-treatment groups were improved, whilst cell viability and the expression of FSHR were significantly increased but oxidative stress injury was decreased. The present results suggest that L-carnitine can protect the cells from OS damage induced by H2O2, enhance antioxidant activity whilst suppressing the apoptosis of GCs, in addition to preserving FSHR expression in GCs under OS. Therefore, the present study revealed that the introduction of L-carnitine in clinical medicine or dietary supplement may protect GCs, improve follicular quality and female reproductive function.
Keywords: L-carnitine; follicular stimulating hormone receptor; mitochondria; ovarian granulosa cells; oxidative stress.
Copyright: © Li et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
TRIB3 regulates FSHR expression in human granulosa cells under high levels of free fatty acids.Reprod Biol Endocrinol. 2021 Sep 9;19(1):139. doi: 10.1186/s12958-021-00823-z. Reprod Biol Endocrinol. 2021. PMID: 34503515 Free PMC article.
-
Protective effect of afamin protein against oxidative stress related injury in human ovarian granulosa cells.J Ovarian Res. 2024 Sep 28;17(1):189. doi: 10.1186/s13048-024-01511-3. J Ovarian Res. 2024. PMID: 39342320 Free PMC article.
-
Catalpol protects rat ovarian granulosa cells against oxidative stress and apoptosis through modulating the PI3K/Akt/mTOR signaling pathway.Biosci Rep. 2020 Apr 30;40(4):BSR20194032. doi: 10.1042/BSR20194032. Biosci Rep. 2020. PMID: 32227125 Free PMC article.
-
Oxidative Stress in Granulosa-Lutein Cells From In Vitro Fertilization Patients.Reprod Sci. 2016 Dec;23(12):1656-1661. doi: 10.1177/1933719116674077. Reprod Sci. 2016. PMID: 27821562 Review.
-
Oxidative stress and mitochondrial dysfunction of granulosa cells in polycystic ovarian syndrome.Front Med (Lausanne). 2023 Jun 28;10:1193749. doi: 10.3389/fmed.2023.1193749. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37448805 Free PMC article. Review.
Cited by
-
The effect of L-carnitine in reactive oxygen species reduction and apoptotic gene expression in mice after cyclophosphamide: An experimental study.Int J Reprod Biomed. 2024 Oct 14;22(8):661-672. doi: 10.18502/ijrm.v22i8.17262. eCollection 2024 Aug. Int J Reprod Biomed. 2024. PMID: 39494123 Free PMC article.
-
In vitro evidence of antioxidant and anti-inflammatory effects of a new nutraceutical formulation explains benefits in a clinical setting of COPD patients.Front Pharmacol. 2024 Aug 20;15:1439835. doi: 10.3389/fphar.2024.1439835. eCollection 2024. Front Pharmacol. 2024. PMID: 39228520 Free PMC article.
References
-
- Zhang H, Luo Q, Lu X, Yin N, Zhou D, Zhang L, Zhao W, Wang D, Du P, Hou Y, et al. Effects of hPMSCs on granulosa cell apoptosis and AMH expression and their role in the restoration of ovary function in premature ovarian failure mice. Stem Cell Res Ther. 2018;9(20) doi: 10.1186/s13287-017-0745-5. - DOI - PMC - PubMed
-
- Terao H, Wada-Hiraike O, Nagumo A, Kunitomi C, Azhary JMK, Harada M, Hirata T, Hirota Y, Koga K, Fujii T, Osuga Y. Role of oxidative stress in follicular fluid on embryos of patients undergoing assisted reproductive technology treatment. J Obstet Gynaecol Res. 2019;45:1884–1891. doi: 10.1111/jog.14040. - DOI - PubMed
LinkOut - more resources
Full Text Sources
