Early Endosomes Act as Local Exocytosis Hubs to Repair Endothelial Membrane Damage
- PMID: 36938863
- PMCID: PMC10161044
- DOI: 10.1002/advs.202300244
Early Endosomes Act as Local Exocytosis Hubs to Repair Endothelial Membrane Damage
Abstract
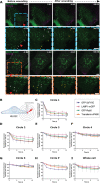
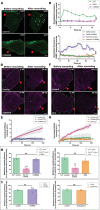
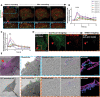
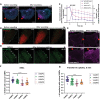
The plasma membrane of a cell is subject to stresses causing ruptures that must be repaired immediately to preserve membrane integrity and ensure cell survival. Yet, the spatio-temporal membrane dynamics at the wound site and the source of the membrane required for wound repair are poorly understood. Here, it is shown that early endosomes, previously only known to function in the uptake of extracellular material and its endocytic transport, are involved in plasma membrane repair in human endothelial cells. Using live-cell imaging and correlative light and electron microscopy, it is demonstrated that membrane injury triggers a previously unknown exocytosis of early endosomes that is induced by Ca2+ entering through the wound. This exocytosis is restricted to the vicinity of the wound site and mediated by the endosomal soluble N-ethylmaleimide sensitive factor attachment protein receptor (SNARE) VAMP2, which is crucial for efficient membrane repair. Thus, the newly identified Ca2+ -evoked and localized exocytosis of early endosomes supplies the membrane material required for rapid resealing of a damaged plasma membrane, thereby providing the first line of defense against damage in mechanically challenged endothelial cells.
Keywords: VAMP2; calcium; endosomal fusion; membrane wounds; plasma membrane resealing.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
