Podocyte protease activated receptor 1 stimulation in mice produces focal segmental glomerulosclerosis mirroring human disease signaling events
- PMID: 36940798
- PMCID: PMC7616342
- DOI: 10.1016/j.kint.2023.02.031
Podocyte protease activated receptor 1 stimulation in mice produces focal segmental glomerulosclerosis mirroring human disease signaling events
Abstract
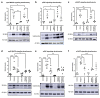
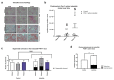
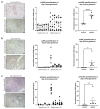
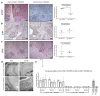
About 30% of patients who have a kidney transplant with underlying nephrotic syndrome (NS) experience rapid relapse of disease in their new graft. This is speculated to be due to a host-derived circulating factor acting on podocytes, the target cells in the kidney, leading to focal segmental glomerulosclerosis (FSGS). Our previous work suggests that podocyte membrane protease receptor 1 (PAR-1) is activated by a circulating factor in relapsing FSGS. Here, the role of PAR-1 was studied in human podocytes in vitro, and using a mouse model with developmental or inducible expression of podocyte-specific constitutively active PAR-1, and using biopsies from patients with nephrotic syndrome. In vitro podocyte PAR-1 activation caused a pro-migratory phenotype with phosphorylation of the kinase JNK, VASP protein and docking protein Paxillin. This signaling was mirrored in podocytes exposed to patient relapse-derived NS plasma and in patient disease biopsies. Both developmental and inducible activation of transgenic PAR-1 (NPHS2 Cre PAR-1Active+/-) caused early severe nephrotic syndrome, FSGS, kidney failure and, in the developmental model, premature death. We found that the non-selective cation channel protein TRPC6 could be a key modulator of PAR-1 signaling and TRPC6 knockout in our mouse model significantly improved proteinuria and extended lifespan. Thus, our work implicates podocyte PAR-1 activation as a key initiator of human NS circulating factor and that the PAR-1 signaling effects were partly modulated through TRPC6.
Keywords: PAR-1; circulating factor; nephrotic syndrome; podocyte; proteases.
Crown Copyright © 2023. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
All the authors declared no competing interests.
Figures




Comment in
-
Stage lighting on PAR-1: a step further in the understanding of acquired focal and segmental glomerulosclerosis.Kidney Int. 2023 Aug;104(2):234-236. doi: 10.1016/j.kint.2023.04.016. Kidney Int. 2023. PMID: 37479383
References
-
- Harris JJ, McCarthy HJ, Ni L, et al. Active proteases in nephrotic plasma lead to a podocin-dependent phosphorylation of VASP in podocytes via protease activated receptor-1. J Pathol. 2013;229:660–671. - PubMed
-
- Bierzynska A, McCarthy HJ, Soderquest K, et al. Genomic and clinical profiling of a national nephrotic syndrome cohort advocates a precision medicine approach to disease management. Kidney Int. 2017;91:937–947. - PubMed
-
- Oleinika K, Mauri C, Salama AD. Effector and regulatory B cells in immune-mediated kidney disease. Nat Rev Nephrol. 2019;15:11–26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

