Gut microbe-derived milnacipran enhances tolerance to gut ischemia/reperfusion injury
- PMID: 36948152
- PMCID: PMC10040455
- DOI: 10.1016/j.xcrm.2023.100979
Gut microbe-derived milnacipran enhances tolerance to gut ischemia/reperfusion injury
Abstract
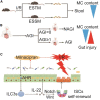
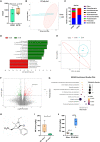
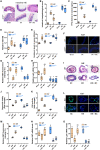
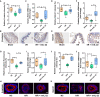
There are significant differences in the susceptibility of populations to intestinal ischemia/reperfusion (I/R), but the underlying mechanisms remain elusive. Here, we show that mice exhibit significant differences in susceptibility to I/R-induced enterogenic sepsis. Notably, the milnacipran (MC) content in the enterogenic-sepsis-tolerant mice is significantly higher. We also reveal that the pre-operative fecal MC content in cardiopulmonary bypass patients, including those with intestinal I/R injury, is associated with susceptibility to post-operative gastrointestinal injury. We reveal that MC attenuates mouse I/R injury in wild-type mice but not in intestinal epithelial aryl hydrocarbon receptor (AHR) gene conditional knockout mice (AHRflox/flox) or IL-22 gene deletion mice (IL-22-/-). Collectively, our results suggest that gut microbiota affects susceptibility to I/R-induced enterogenic sepsis and that gut microbiota-derived MC plays a pivotal role in tolerance to intestinal I/R in an AHR/ILC3/IL-22 signaling-dependent manner, revealing the pathological mechanism, potential prevention and treatment drugs, and treatment strategies for intestinal I/R.
Keywords: IL-22; aryl hydrocarbon receptor; enterogenic sepsis; intestinal flora; milnacipran.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare that they have no conflict of interests.
Figures




References
-
- Deng F., Zhao B.C., Yang X., Lin Z.B., Sun Q.S., Wang Y.F., Yan Z.Z., Liu W.F., Li C., Hu J.J., Liu K.X. The gut microbiota metabolite capsiate promotes Gpx4 expression by activating TRPV1 to inhibit intestinal ischemia reperfusion-induced ferroptosis. Gut Microb. 2021;13:1–21. doi: 10.1080/19490976.2021.1902719. - DOI - PMC - PubMed
-
- Singer M., Deutschman C.S., Seymour C.W., Shankar-Hari M., Annane D., Bauer M., Bellomo R., Bernard G.R., Chiche J.D., Coopersmith C.M., et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:801–810. doi: 10.1001/jama.2016.0287. - DOI - PMC - PubMed
-
- Rudd K.E., Johnson S.C., Agesa K.M., Shackelford K.A., Tsoi D., Kievlan D.R., Colombara D.V., Ikuta K.S., Kissoon N., Finfer S., et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet (London, England) 2020;395:200–211. doi: 10.1016/s0140-6736(19)32989-7. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

