Impact of additional genetic abnormalities at diagnosis of chronic myeloid leukemia for first-line imatinib-treated patients receiving proactive treatment intervention
- PMID: 36951160
- PMCID: PMC10483360
- DOI: 10.3324/haematol.2022.282184
Impact of additional genetic abnormalities at diagnosis of chronic myeloid leukemia for first-line imatinib-treated patients receiving proactive treatment intervention
Abstract
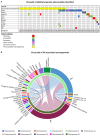
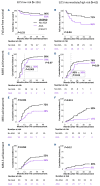
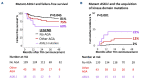
The BCR::ABL1 gene fusion initiates chronic myeloid leukemia (CML); however, evidence has accumulated from studies of highly selected cohorts that variants in other cancer-related genes are associated with treatment failure. Nevertheless, the true incidence and impact of additional genetic abnormalities (AGA) at diagnosis of chronic phase (CP)-CML is unknown. We sought to determine whether AGA at diagnosis in a consecutive imatinib-treated cohort of 210 patients enrolled in the TIDEL-II trial influenced outcome despite a highly proactive treatment intervention strategy. Survival outcomes including overall survival, progression-free survival, failure-free survival, and BCR::ABL1 kinase domain mutation acquisition were evaluated. Molecular outcomes were measured at a central laboratory and included major molecular response (MMR, BCR::ABL1 ≤0.1%IS), MR4 (BCR::ABL1 ≤0.01%IS), and MR4.5 (BCR::ABL1 ≤0.0032%IS). AGA included variants in known cancer genes and novel rearrangements involving the formation of the Philadelphia chromosome. Clinical outcomes and molecular response were assessed based on the patient's genetic profile and other baseline factors. AGA were identified in 31% of patients. Potentially pathogenic variants in cancer-related genes were detected in 16% of patients at diagnosis (including gene fusions and deletions) and structural rearrangements involving the Philadelphia chromosome (Ph-associated rearrangements) were detected in 18%. Multivariable analysis demonstrated that the combined genetic abnormalities plus the EUTOS long-term survival clinical risk score were independent predictors of lower molecular response rates and higher treatment failure. Despite a highly proactive treatment intervention strategy, first-line imatinib-treated patients with AGA had poorer response rates. These data provide evidence for the incorporation of genomically-based risk assessment for CML.
Figures



Comment in
-
New patterns of genetic instability in chronic myeloid leukemia: interesting, but not ready for clinical use.Haematologica. 2023 Sep 1;108(9):2273-2274. doi: 10.3324/haematol.2023.283059. Haematologica. 2023. PMID: 37078263 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

