Molecular targeting of the UDP-glucuronosyltransferase enzymes in high-eukaryotic translation initiation factor 4E refractory/relapsed acute myeloid leukemia patients: a randomized phase II trial of vismodegib, ribavirin with or without decitabine
- PMID: 36951168
- PMCID: PMC10620574
- DOI: 10.3324/haematol.2023.282791
Molecular targeting of the UDP-glucuronosyltransferase enzymes in high-eukaryotic translation initiation factor 4E refractory/relapsed acute myeloid leukemia patients: a randomized phase II trial of vismodegib, ribavirin with or without decitabine
Abstract
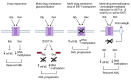
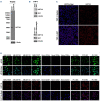
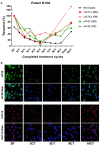
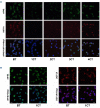
Drug resistance underpins poor outcomes in many malignancies including refractory and relapsed acute myeloid leukemia (R/R AML). Glucuronidation is a common mechanism of drug inactivation impacting many AML therapies, e.g., cytarabine, decitabine, azacytidine and venetoclax. In AML cells, the capacity for glucuronidation arises from increased production of the UDP-glucuronosyltransferase 1A (UGT1A) enzymes. UGT1A elevation was first observed in AML patients who relapsed after response to ribavirin, a drug used to target the eukaryotic translation initiation factor eIF4E, and subsequently in patients who relapsed on cytarabine. UGT1A elevation resulted from increased expression of the sonic-hedgehog transcription factor GLI1. Vismodegib inhibited GLI1, decreased UGT1A levels, reduced glucuronidation of ribavirin and cytarabine, and re-sensitized cells to these drugs. Here, we examined if UGT1A protein levels, and thus glucuronidation activity, were targetable in humans and if this corresponded to clinical response. We conducted a phase II trial using vismodegib with ribavirin, with or without decitabine, in largely heavily pre-treated patients with high-eIF4E AML. Pre-therapy molecular assessment of patients' blasts indicated highly elevated UGT1A levels relative to healthy volunteers. Among patients with partial response, blast response or prolonged stable disease, vismodegib reduced UGT1A levels, which corresponded to effective targeting of eIF4E by ribavirin. In all, our studies are the first to demonstrate that UGT1A protein, and thus glucuronidation, are targetable in humans. These studies pave the way for the development of therapies that impair glucuronidation, one of the most common drug deactivation modalities. Clinicaltrials.gov: NCT02073838.
Figures
Comment in
-
A novel approach to overcome drug resistance in acute myeloid leukemia.Haematologica. 2023 Nov 1;108(11):2889-2890. doi: 10.3324/haematol.2023.283099. Haematologica. 2023. PMID: 37165841 Free PMC article. No abstract available.
Similar articles
-
The sonic hedgehog factor GLI1 imparts drug resistance through inducible glucuronidation.Nature. 2014 Jul 3;511(7507):90-3. doi: 10.1038/nature13283. Epub 2014 May 28. Nature. 2014. PMID: 24870236 Free PMC article.
-
When will resistance be futile?Cancer Res. 2014 Dec 15;74(24):7175-80. doi: 10.1158/0008-5472.CAN-14-2607. Epub 2014 Dec 4. Cancer Res. 2014. PMID: 25477336 Free PMC article. Review.
-
Molecular targeting of the oncogene eIF4E in acute myeloid leukemia (AML): a proof-of-principle clinical trial with ribavirin.Blood. 2009 Jul 9;114(2):257-60. doi: 10.1182/blood-2009-02-205153. Epub 2009 May 11. Blood. 2009. PMID: 19433856 Clinical Trial.
-
A phase I trial of ribavirin and low-dose cytarabine for the treatment of relapsed and refractory acute myeloid leukemia with elevated eIF4E.Haematologica. 2015 Jan;100(1):e7-9. doi: 10.3324/haematol.2014.111245. Epub 2014 Nov 25. Haematologica. 2015. PMID: 25425688 Free PMC article. Clinical Trial. No abstract available.
-
An evaluation of venetoclax in combination with azacitidine, decitabine, or low-dose cytarabine as therapy for acute myeloid leukemia.Expert Rev Hematol. 2021 May;14(5):407-417. doi: 10.1080/17474086.2021.1938533. Epub 2021 Jun 15. Expert Rev Hematol. 2021. PMID: 34076549 Free PMC article. Review.
Cited by
-
eIF4E orchestrates mRNA processing, RNA export and translation to modify specific protein production.Nucleus. 2024 Dec;15(1):2360196. doi: 10.1080/19491034.2024.2360196. Epub 2024 Jun 16. Nucleus. 2024. PMID: 38880976 Free PMC article. Review.
-
Multipurpose RNA maturation factors dysregulate multiple mRNA processing steps simultaneously and provide new therapeutic opportunities.RNA Biol. 2025 Dec;22(1):1-14. doi: 10.1080/15476286.2025.2503040. Epub 2025 Jun 9. RNA Biol. 2025. PMID: 40485569 Free PMC article. Review.
-
A novel approach to overcome drug resistance in acute myeloid leukemia.Haematologica. 2023 Nov 1;108(11):2889-2890. doi: 10.3324/haematol.2023.283099. Haematologica. 2023. PMID: 37165841 Free PMC article. No abstract available.
-
Posttranscriptional activity of the eukaryotic translation initiation factor eIF4E contributes to HoxA9-driven leukemogenesis.bioRxiv [Preprint]. 2025 Feb 12:2025.02.10.637540. doi: 10.1101/2025.02.10.637540. bioRxiv. 2025. PMID: 39990322 Free PMC article. Preprint.
-
The eukaryotic translation initiation factor eIF4E unexpectedly acts in splicing thereby coupling mRNA processing with translation: eIF4E induces widescale splicing reprogramming providing system-wide connectivity between splicing, nuclear mRNA export and translation.Bioessays. 2024 Jan;46(1):e2300145. doi: 10.1002/bies.202300145. Epub 2023 Nov 5. Bioessays. 2024. PMID: 37926700 Free PMC article.
References
-
- de Lima M, Roboz GJ, Platzbecker U, Craddock C, Ossenkoppele G. AML and the art of remission maintenance. Blood Rev. 2021;49:100829. - PubMed
-
- DiNardo CD, Rausch CR, Benton C, et al. . Clinical experience with the BCL2-inhibitor venetoclax in combination therapy for relapsed and refractory acute myeloid leukemia and related myeloid malignancies. Am J Hematol. 2018;93(3):401-407. - PubMed
-
- DiNardo CD, Jonas BA, Pullarkat V, et al. . Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383(7):617-629. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

