Local administration of mesenchymal stromal cells is safe and modulates the immune compartment in ulcerative proctitis
- PMID: 36951952
- PMCID: PMC10243829
- DOI: 10.1172/jci.insight.167402
Local administration of mesenchymal stromal cells is safe and modulates the immune compartment in ulcerative proctitis
Abstract
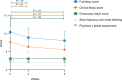
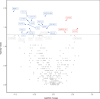
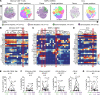
BACKGROUNDDue to their immunoregulatory and tissue regenerative features, mesenchymal stromal cells (MSCs) are a promising novel tool for the management of ulcerative proctitis (UP). Here we report on a phase IIa clinical study that evaluated the impact of local MSC therapy on UP.METHODSThirteen refractory UP patients, with an endoscopic Mayo score (EMS) of 2 or 3, were included. Seven patients received 20-40 million allogeneic MSCs (cohort 1), while 6 patients received 40-80 million MSCs (cohort 2). Adverse events (AEs) were assessed at baseline and on weeks 2, 6, 12, and 24. Clinical, endoscopic, and biochemical parameters were assessed at baseline and on weeks 2 and 6. Furthermore, we evaluated the engraftment of MSCs, the presence of donor-specific human leukocyte antigen (HLA) antibodies (DSAs), and we determined the impact of MSC therapy on the local immune compartment.RESULTSNo serious AEs were observed. The clinical Mayo score was significantly improved on weeks 2 and 6, and the EMS was significantly improved on week 6, compared with baseline. On week 6, donor MSCs were still detectable in rectal biopsies from 4 of 9 patients and DSAs against both HLA class I and class II were found. Mass cytometry showed a reduction in activated CD8+ T cells and CD16+ monocytes and an enrichment in mononuclear phagocytes and natural killer cells in biopsies after local MSC therapy.CONCLUSIONLocal administration of allogeneic MSCs is safe, tolerable, and feasible for treatment of refractory UP and shows encouraging signs of clinical efficacy and modulation of local immune responses. This sets the stage for larger clinical trials.TRIAL REGISTRATIONEU Clinical Trials Register (EudraCT, 2017-003524-75) and the Dutch Trial Register (NTR7205).FUNDINGECCO grant 2020.
Keywords: Clinical Trials; Inflammation; Inflammatory bowel disease.
Figures



References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

