Tumor-associated macrophages respond to chemotherapy by detrimental transcriptional reprogramming and suppressing stabilin-1 mediated clearance of EGF
- PMID: 36960065
- PMCID: PMC10028613
- DOI: 10.3389/fimmu.2023.1000497
Tumor-associated macrophages respond to chemotherapy by detrimental transcriptional reprogramming and suppressing stabilin-1 mediated clearance of EGF
Abstract
Introduction: Tumor resistance to chemotherapy and metastatic relapse account for more than 90% of cancer specific mortality. Tumor-associated macrophages (TAMs) can process chemotherapeutic agents and impair their action. Little is known about the direct effects of chemotherapy on TAMs.
Methods: The effect of chemotherapeutic platinum agent cisplatin was assessed in the model system of human ex vivo TAMs. Whole-transcriptome sequencing for paired TAMs stimulated and not stimulated by cisplatin was analysed by NGS. Endocytic uptake of EGF was quantified by flow cytometry. Confocal microscopy was used to visualize stabilin-1-mediated internalization and endocytic trafficking of EGF in CHO cells expressing ectopically recombinant stabilin-1 and in stabilin-1+ TAMs. In cohort of patients with breast cancer, the effect of platinum therapy on the transcriptome of TAMs was validated, and differential expression of regulators of endocytosis was identified.
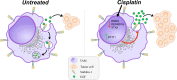
Results: Here we show that chemotherapeutic agent cisplatin can initiate detrimental transcriptional and functional programs in TAMs, without significant impairment of their viability. We focused on the clearance function of TAMs that controls composition of tumor microenvironment. For the first time we demonstrated that TAMs' scavenger receptor stabilin-1 is responsible for the clearance of epidermal growth factor (EGF), a potent stimulator of tumor growth. Cisplatin suppressed both overall and EGF-specific endocytosis in TAMs by bidirectional mode: suppression of positive regulators and stimulation of negative regulators of endocytosis, with strongest effect on synaptotagmin-11 (SYT11), confirmed in patients with breast cancer.
Conclusion: Our data demonstrate that synergistic action of cytostatic agents and innovative immunomodulators is required to overcome cancer therapy resistance.
Keywords: EGF; breast cancer; cisplatin; clearance; endocytosis; stabilin-1; tumor-associated macrophage (TAM).
Copyright © 2023 Larionova, Kiselev, Kazakova, Liu, Patysheva, Iamshchikov, Liu, Mossel, Riabov, Rakina, Sergushichev, Bezgodova, Vtorushin, Litviakov, Denisov, Koshkin, Pyankov, Tsyganov, Ibragimova, Cherdyntseva and Kzhyshkowska.
Conflict of interest statement
Authors DP and PK were employed by company Genomed. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Stabilin-1 is expressed in human breast cancer and supports tumor growth in mammary adenocarcinoma mouse model.Oncotarget. 2016 May 24;7(21):31097-110. doi: 10.18632/oncotarget.8857. Oncotarget. 2016. PMID: 27105498 Free PMC article.
-
Titanium Nanoparticles Enhance Production and Suppress Stabilin-1-Mediated Clearance of GDF-15 in Human Primary Macrophages.Front Immunol. 2021 Dec 15;12:760577. doi: 10.3389/fimmu.2021.760577. eCollection 2021. Front Immunol. 2021. PMID: 34975851 Free PMC article.
-
Novel function of alternatively activated macrophages: stabilin-1-mediated clearance of SPARC.J Immunol. 2006 May 15;176(10):5825-32. doi: 10.4049/jimmunol.176.10.5825. J Immunol. 2006. PMID: 16670288
-
Tumor-Associated Macrophages: Critical Players in Drug Resistance of Breast Cancer.Front Immunol. 2021 Dec 17;12:799428. doi: 10.3389/fimmu.2021.799428. eCollection 2021. Front Immunol. 2021. PMID: 34992609 Free PMC article. Review.
-
Tumor-Associated Macrophages in Human Breast, Colorectal, Lung, Ovarian and Prostate Cancers.Front Oncol. 2020 Oct 22;10:566511. doi: 10.3389/fonc.2020.566511. eCollection 2020. Front Oncol. 2020. PMID: 33194645 Free PMC article. Review.
Cited by
-
Modelling of macrophage responses to biomaterials in vitro: state-of-the-art and the need for the improvement.Front Immunol. 2024 Mar 26;15:1349461. doi: 10.3389/fimmu.2024.1349461. eCollection 2024. Front Immunol. 2024. PMID: 38596667 Free PMC article. Review.
-
The role of cisplatin in modulating the tumor immune microenvironment and its combination therapy strategies: a new approach to enhance anti-tumor efficacy.Ann Med. 2025 Dec;57(1):2447403. doi: 10.1080/07853890.2024.2447403. Epub 2025 Jan 6. Ann Med. 2025. PMID: 39757995 Free PMC article. Review.
-
Chemotherapy Enriches for Proinflammatory Macrophage Phenotypes that Support Cancer Stem-Like Cells and Disease Progression in Ovarian Cancer.Cancer Res Commun. 2024 Oct 1;4(10):2638-2652. doi: 10.1158/2767-9764.CRC-24-0311. Cancer Res Commun. 2024. PMID: 39287565 Free PMC article.
-
Targeting of TAMs: can we be more clever than cancer cells?Cell Mol Immunol. 2024 Dec;21(12):1376-1409. doi: 10.1038/s41423-024-01232-z. Epub 2024 Nov 8. Cell Mol Immunol. 2024. PMID: 39516356 Free PMC article. Review.
-
Macrophages and platelets in liver fibrosis and hepatocellular carcinoma.Front Immunol. 2023 Dec 5;14:1277808. doi: 10.3389/fimmu.2023.1277808. eCollection 2023. Front Immunol. 2023. PMID: 38116017 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

