Region-specific laboratory reference intervals are important: A systematic review of the data from Africa
- PMID: 36962599
- PMCID: PMC10021479
- DOI: 10.1371/journal.pgph.0000783
Region-specific laboratory reference intervals are important: A systematic review of the data from Africa
Abstract
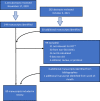
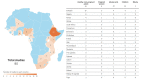
Region-specific laboratory reference intervals (RIs) are important for clinical trials and these data are often sparse in priority areas for research, including Africa. We reviewed data on RIs from Africa to identify gaps in the literature with a systematic review of PubMed for RI studies from Africa published ≥2010. Search focus included clinical analytic chemistry, hematology, immunological parameters and RIs. Data from adults, adolescents, children, pregnant women, and the elderly were included. We excluded manuscripts reporting data from persons with conditions that might preclude clinical trial participation in studies enrolling healthy volunteers. Of 179 identified manuscripts, 80 were included in this review, covering 20 countries with the largest number of studies in Ethiopia (n = 23, 29%). Most studies considered healthy, nonpregnant adults (n = 55, 69%). Nine (11%) studies included pregnant women, 13 (16%) included adolescents and 22 (28%) included children. Recruitment, screening, enrollment procedures and definition of age strata varied across studies. The most common type of RIs reported were hematology (66, 83%); 14 studies (18%) included flow cytometry and/or T cell counts. Other common tests or panels included liver function assays (32, 40%), renal function assays (30, 38%), lipid chemistries (17, 21%) and serum electrolytes (17, 21%). The number of parameters characterized ranged from only one (three studies characterized either CD4+ counts, D-dimer, or hemoglobin), to as many as 40. Statistical methods for calculating RIs varied. 56 (70%) studies compared their results to international RI databases. Though most presented their data side-by-side with international data with little accompanying analysis, nearly all reported deviation from comparator RI data, sometimes with half or more of otherwise healthy participants having an "out of range" result. We found there is limited local RI data available in sub-Saharan Africa. Studies to fill this gap are warranted, including efforts to standardize statistical methods to derive RIs, methods to compare with other RIs, and improve representative participant selection.
Copyright: © 2022 Price et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- CLSI. Defining, Establishing and Verifying Reference Intervals in the Clinical Laboratory: Approved Guidelines—Third Edition. Wayne, PA, USA: Clinical and Laboratory Standards Institute; 2008.
-
- IFCC/C-RIDL. [14 Apr 2021]. Available from: https://www.ifcc.org/ifcc-scientific-division/sd-committees/c-ridl/.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
