Continuous Measurement of Lactate Concentration in Human Subjects through Direct Electron Transfer from Enzymes to Microneedle Electrodes
- PMID: 36967522
- PMCID: PMC10152478
- DOI: 10.1021/acssensors.2c02780
Continuous Measurement of Lactate Concentration in Human Subjects through Direct Electron Transfer from Enzymes to Microneedle Electrodes
Abstract
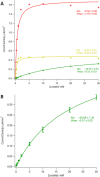
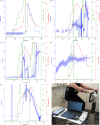
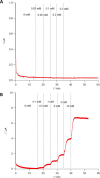
Microneedle lactate sensors may be used to continuously measure lactate concentration in the interstitial fluid in a minimally invasive and pain-free manner. First- and second-generation enzymatic sensors produce a redox-active product that is electrochemically sensed at the electrode surface. Direct electron transfer enzymes produce electrons directly as the product of enzymatic action; in this study, a direct electron transfer enzyme specific to lactate has been immobilized onto a microneedle surface to create lactate-sensing devices that function at low applied voltages (0.2 V). These devices have been validated in a small study of human volunteers; lactate concentrations were raised and lowered through physical exercise and subsequent rest. Lactazyme microneedle devices show good agreement with concurrently obtained and analyzed serum lactate levels.
Keywords: biosensor; continuous monitoring; direct electron transfer enzyme; in-human study; lactate; microneedles.
Conflict of interest statement
The authors declare the following competing financial interest(s): AEGC is the founder of a company, Continuous Diagnostics Ltd., involved in commercialisation of microneedle sensing devices. AKGF is the co-founder of a company, DirectSens GmbH, involved in commercialisation of enzyme-based sensor technology.
Figures

Similar articles
-
Microneedle-based biosensor for minimally-invasive lactate detection.Biosens Bioelectron. 2019 Jan 1;123:152-159. doi: 10.1016/j.bios.2018.08.010. Epub 2018 Aug 10. Biosens Bioelectron. 2019. PMID: 30177422
-
Reduced graphene oxide-functionalized polymer microneedle for continuous and wide-range monitoring of lactate in interstitial fluid.Talanta. 2024 Apr 1;270:125582. doi: 10.1016/j.talanta.2023.125582. Epub 2023 Dec 21. Talanta. 2024. PMID: 38176248
-
Continuous Lactate Monitoring System Based on Percutaneous Microneedle Array.Sensors (Basel). 2022 Feb 14;22(4):1468. doi: 10.3390/s22041468. Sensors (Basel). 2022. PMID: 35214368 Free PMC article.
-
Tackling the challenges of developing microneedle-based electrochemical sensors.Mikrochim Acta. 2022 Nov 3;189(11):440. doi: 10.1007/s00604-022-05510-3. Mikrochim Acta. 2022. PMID: 36329339 Review.
-
Electron Transfer Between Enzymes and Electrodes.Adv Biochem Eng Biotechnol. 2019;167:39-85. doi: 10.1007/10_2017_42. Adv Biochem Eng Biotechnol. 2019. PMID: 29224083 Review.
Cited by
-
Lateral Flow-Based Skin Patch for Rapid Detection of Protein Biomarkers in Human Dermal Interstitial Fluid.ACS Sens. 2024 Nov 22;9(11):5792-5801. doi: 10.1021/acssensors.4c00956. Epub 2024 Oct 25. ACS Sens. 2024. PMID: 39455057 Free PMC article.
-
Microneedle sensors for dermal interstitial fluid analysis.Med X. 2024;2(1):15. doi: 10.1007/s44258-024-00028-0. Epub 2024 Oct 1. Med X. 2024. PMID: 39363915 Free PMC article. Review.
-
Recent Advances in Amperometric Biosensors for Medical Applications: A Mini-Review.Curr Top Med Chem. 2025;25(7):741-760. doi: 10.2174/0115680266323004241015122441. Curr Top Med Chem. 2025. PMID: 39473247 Review.
-
Can we still use the Michaelis-Menten model for enzymatic microneedle sensors?Proc Natl Acad Sci U S A. 2025 Jul 29;122(30):e2418168122. doi: 10.1073/pnas.2418168122. Epub 2025 Jul 23. Proc Natl Acad Sci U S A. 2025. PMID: 40699925
-
Self-Extracting Dextran-Based Hydrogel Microneedle Arrays with an Interpenetrating Bioelectroenzymatic Sensor for Transdermal Monitoring with Matrix Protection.Adv Healthc Mater. 2025 Jan;14(2):e2403209. doi: 10.1002/adhm.202403209. Epub 2024 Nov 24. Adv Healthc Mater. 2025. PMID: 39580665 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

