Prevention strategies of postoperative adhesion in soft tissues by applying biomaterials: Based on the mechanisms of occurrence and development of adhesions
- PMID: 36969107
- PMCID: PMC10030827
- DOI: 10.1016/j.bioactmat.2023.02.026
Prevention strategies of postoperative adhesion in soft tissues by applying biomaterials: Based on the mechanisms of occurrence and development of adhesions
Abstract
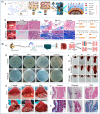
Postoperative adhesion (POA) widely occurs in soft tissues and usually leads to chronic pain, dysfunction of adjacent organs and some acute complications, seriously reducing patients' quality of life and even being life-threatening. Except for adhesiolysis, there are few effective methods to release existing adhesion. However, it requires a second operation and inpatient care and usually triggers recurrent adhesion in a great incidence. Hence, preventing POA formation has been regarded as the most effective clinical strategy. Biomaterials have attracted great attention in preventing POA because they can act as both barriers and drug carriers. Nevertheless, even though much reported research has been demonstrated their efficacy on POA inhibition to a certain extent, thoroughly preventing POA formation is still challenging. Meanwhile, most biomaterials for POA prevention were designed based on limited experiences, not a solid theoretical basis, showing blindness. Hence, we aimed to provide guidance for designing anti-adhesion materials applied in different soft tissues based on the mechanisms of POA occurrence and development. We first classified the postoperative adhesions into four categories according to the different components of diverse adhesion tissues, and named them as "membranous adhesion", "vascular adhesion", "adhesive adhesion" and "scarred adhesion", respectively. Then, the process of the occurrence and development of POA were analyzed, and the main influencing factors in different stages were clarified. Further, we proposed seven strategies for POA prevention by using biomaterials according to these influencing factors. Meanwhile, the relevant practices were summarized according to the corresponding strategies and the future perspectives were analyzed.
Keywords: Biomaterials; Mechanisms of occurrence and development; Prevention of postoperative adhesions (POA); Soft tissues.
© 2023 The Authors.
Conflict of interest statement
There are no conflicts of interest to declare.
Figures





References
-
- Foster D.S., Marshall C.D., Gulati G.S., Chinta M.S., Nguyen A., Salhotra A., Jones R.E., Burcham A., Lerbs T., Cui L., King M.E., Titan A.L., Ransom R.C., Manjunath A., Hu M.S., Blackshear C.P., Mascharak S., Moore A.L., Norton J.A., Kin C.J., Shelton A.A., Januszyk M., Gurtner G.C., Wernig G., Longaker M.T. Elucidating the fundamental fibrotic processes driving abdominal adhesion formation. Nat. Commun. 2020;11:4061. - PMC - PubMed
-
- Okabayashi K., Ashrafian H., Zacharakis E., Hasegawa H., Kitagawa Y., Athanasiou T., Darzi A. Adhesions after abdominal surgery: a systematic review of the incidence, distribution and severity. Surg. Today. 2014;44:405–420. - PubMed
-
- Brochhausen C., Schmitt V.H., Planck C.N.E., Rajab T.K., Hollemann D., Tapprich C., Krämer B., Wallwiener C., Hierlemann H., Zehbe R., Planck H., Kirkpatrick C.J. Current strategies and future perspectives for intraperitoneal adhesion prevention. J. Gastrointest. Surg. 2012;16:1256–1274. - PubMed
-
- Yang J., Bai R., Chen B., Suo Z. Hydrogel adhesion: a supramolecular synergy of chemistry, topology, and mechanics. Adv. Funct. Mater. 2020;30
Publication types
LinkOut - more resources
Full Text Sources

