Stage migration as a surrogate of survival in hepatocellular carcinoma treated with transarterial chemoembolization
- PMID: 36972387
- PMCID: PMC10043575
- DOI: 10.1097/HC9.0000000000000091
Stage migration as a surrogate of survival in hepatocellular carcinoma treated with transarterial chemoembolization
Abstract
Background aims: Locoregional therapies, including transarterial chemoembolization (TACE), are recommended for the treatment of HCC; however, clinical trials evaluating their effectiveness have been complicated by a lack of validated surrogate outcomes. We aimed to evaluate if stage migration could serve as a potential surrogate of overall survival in patients undergoing TACE.
Approach: We conducted a retrospective cohort study of adult patients with HCC who underwent TACE as initial therapy from 3 centers in the US from 2008 to 2019. The primary outcome was overall survival from the date of the first TACE treatment, and the primary exposure of interest was Barcelona Clinic Liver Cancer stage migration to a more advanced stage within 6 months of TACE. Survival analysis was completed using Kaplan-Meier and multiple Cox proportional hazard models adjusted by the site.
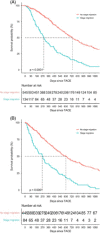
Results: Of 651 eligible patients (51.9% Barcelona Clinic Liver Cancer stage A and 39.6% stage B), 129 (19.6%) patients experienced stage migration within 6 months of TACE. Those with stage migration had larger tumors (5.6 vs. 4.2 cm, p < 0.01) and higher AFP levels (median 92 vs. 15 ng/mL, p < 0.01). In multivariate analysis, stage migration was significantly associated with worse survival (HR: 2.82, 95% CI: 2.66-2.98), with a median survival of 8.7 and 15.9 months in those with and without stage migration. Other predictors of worse survival included the White race, higher AFP levels, a higher number of tumors, and a larger maximum HCC diameter.
Conclusion: Stage migration is associated with increased mortality after TACE in patients with HCC and could serve as a surrogate end point in clinical trials evaluating locoregional therapies such as TACE.
Copyright © 2023 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Association for the Study of Liver Diseases.
Conflict of interest statement
Amit G. Singal served as a consultant or on advisory boards for Genentech, Astra Zeneca, Bayer, Eisai, Exelixis, FujiFilm Medical Sciences, Exact Sciences, Roche, Glycotest, and GRAIL. Neehar D. Parikh served as a consultant for Exact Sciences, Eli Lilly, and Freenome; has served on advisory boards of Genentech, Eisai, Bayer, Exelixis, Wako/Fujifilm; and has received research funding from Bayer, Target RWE, Exact Sciences, Genentech and Glycotest. The remainder authors have no conflicts to report.
Figures
References
-
- Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, et al. . Diagnosis, staging, and management of hepatocellular carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68:723–750. - PubMed
-
- Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301–1314. - PubMed
-
- Forner A, Reig ME, de Lope CR, Bruix J. Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Dis. 2010;30:61–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous