The CLDN5 gene at the blood-brain barrier in health and disease
- PMID: 36978081
- PMCID: PMC10044825
- DOI: 10.1186/s12987-023-00424-5
The CLDN5 gene at the blood-brain barrier in health and disease
Abstract
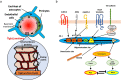
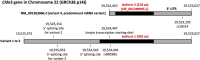
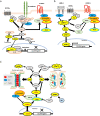
The CLDN5 gene encodes claudin-5 (CLDN-5) that is expressed in endothelial cells and forms tight junctions which limit the passive diffusions of ions and solutes. The blood-brain barrier (BBB), composed of brain microvascular endothelial cells and associated pericytes and end-feet of astrocytes, is a physical and biological barrier to maintain the brain microenvironment. The expression of CLDN-5 is tightly regulated in the BBB by other junctional proteins in endothelial cells and by supports from pericytes and astrocytes. The most recent literature clearly shows a compromised BBB with a decline in CLDN-5 expression increasing the risks of developing neuropsychiatric disorders, epilepsy, brain calcification and dementia. The purpose of this review is to summarize the known diseases associated with CLDN-5 expression and function. In the first part of this review, we highlight the recent understanding of how other junctional proteins as well as pericytes and astrocytes maintain CLDN-5 expression in brain endothelial cells. We detail some drugs that can enhance these supports and are being developed or currently in use to treat diseases associated with CLDN-5 decline. We then summarise mutagenesis-based studies which have facilitated a better understanding of the physiological role of the CLDN-5 protein at the BBB and have demonstrated the functional consequences of a recently identified pathogenic CLDN-5 missense mutation from patients with alternating hemiplegia of childhood. This mutation is the first gain-of-function mutation identified in the CLDN gene family with all others representing loss-of-function mutations resulting in mis-localization of CLDN protein and/or attenuated barrier function. Finally, we summarize recent reports about the dosage-dependent effect of CLDN-5 expression on the development of neurological diseases in mice and discuss what cellular supports for CLDN-5 regulation are compromised in the BBB in human diseases.
Keywords: Blood–brain barrier; Claudin-5; Psychiatric diseases; Tight junction; Vascular permeability.
© 2023. The Author(s).
Conflict of interest statement
Trinity College Dublin owns a patent portfolio related to the use of RNA interference to target claudin-5 for BBB modulation.
Figures

Comment in
-
Genetic disorders and genetic manipulation at the blood-brain barriers.Fluids Barriers CNS. 2023 Apr 20;20(1):29. doi: 10.1186/s12987-023-00428-1. Fluids Barriers CNS. 2023. PMID: 37081497 Free PMC article. No abstract available.
Similar articles
-
The region-selective regulation of endothelial claudin-5 expression and signaling in brain health and disorders.J Cell Physiol. 2021 Oct;236(10):7134-7143. doi: 10.1002/jcp.30357. Epub 2021 Mar 10. J Cell Physiol. 2021. PMID: 33694168 Review.
-
Mosaic deletion of claudin-5 reveals rapid non-cell-autonomous consequences of blood-brain barrier leakage.Cell Rep. 2024 Mar 26;43(3):113911. doi: 10.1016/j.celrep.2024.113911. Epub 2024 Mar 5. Cell Rep. 2024. PMID: 38446668
-
Reversible opening of the blood-brain barrier by claudin-5-binding variants of Clostridium perfringens enterotoxin's claudin-binding domain.Biomaterials. 2018 Apr;161:129-143. doi: 10.1016/j.biomaterials.2018.01.028. Epub 2018 Feb 2. Biomaterials. 2018. PMID: 29421550
-
Recurrent de novo mutations in CLDN5 induce an anion-selective blood-brain barrier and alternating hemiplegia.Brain. 2022 Oct 21;145(10):3374-3382. doi: 10.1093/brain/awac215. Brain. 2022. PMID: 35714222 Free PMC article.
-
Brain endothelial cells and the glio-vascular complex.Cell Tissue Res. 2009 Jan;335(1):75-96. doi: 10.1007/s00441-008-0658-9. Epub 2008 Jul 16. Cell Tissue Res. 2009. PMID: 18633647 Review.
Cited by
-
Genetic disorders and genetic manipulation at the blood-brain barriers.Fluids Barriers CNS. 2023 Apr 20;20(1):29. doi: 10.1186/s12987-023-00428-1. Fluids Barriers CNS. 2023. PMID: 37081497 Free PMC article. No abstract available.
-
Stress-induced mitochondrial fragmentation in endothelial cells disrupts blood-retinal barrier integrity causing neurodegeneration.bioRxiv [Preprint]. 2025 Jan 31:2024.12.21.629919. doi: 10.1101/2024.12.21.629919. bioRxiv. 2025. PMID: 39975311 Free PMC article. Preprint.
-
Biomarkers in Aneurysmatic and Spontaneous Subarachnoid Haemorrhage: A Clinical Prospective Multicentre Biomarker Panel Study of S100B, Claudin-5, Interleukin-10, TREM-1, TREM-2 and Neurofilament Light Chain As Well As Immunoglobulin G and M.Mol Neurobiol. 2025 Sep;62(9):11499-11516. doi: 10.1007/s12035-025-04889-3. Epub 2025 Apr 28. Mol Neurobiol. 2025. PMID: 40295361 Free PMC article.
-
BBB proteomic analysis reveals that complex febrile seizures in infancy enhance susceptibility to epilepsy in adulthood through dysregulation of ECM-receptor interaction signaling pathway.Fluids Barriers CNS. 2025 May 13;22(1):49. doi: 10.1186/s12987-025-00660-x. Fluids Barriers CNS. 2025. PMID: 40361173 Free PMC article.
-
Blood‑brain barrier dysfunction in schizophrenia: Mechanisms and implications (Review).Int J Mol Med. 2025 Oct;56(4):153. doi: 10.3892/ijmm.2025.5594. Epub 2025 Jul 25. Int J Mol Med. 2025. PMID: 40709398 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

