Remotely Programmable Deep Brain Stimulator Combined with an Invasive Blood Pressure Monitoring System for a Non-Tethered Rat Model in Hypertension Research
- PMID: 36979314
- PMCID: PMC10046755
- DOI: 10.3390/brainsci13030504
Remotely Programmable Deep Brain Stimulator Combined with an Invasive Blood Pressure Monitoring System for a Non-Tethered Rat Model in Hypertension Research
Abstract
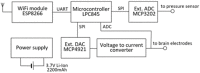
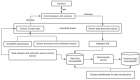
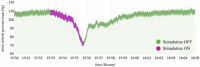
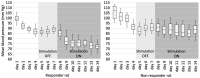
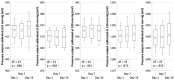
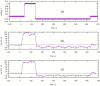
The control circuits of blood pressure have a strong neural regulatory element important in the pathogenesis of essential drug-resistant hypertension. Targeting lower medullary neural control mechanisms of blood pressure by electrical stimulation could be beneficial, and therefore, a novel device is needed. This paper presents a remotely programmable deep brain stimulator with an invasive continuous blood pressure monitoring system in a non-tethered rat model. The device is designed for lower medullary deep brain stimulation research with minimal interference to a daily animal routine. Electrodes were implanted in the caudal ventrolateral medulla. Animal survivability, catheter patency rates, and device data drift were evaluated. Eight out of ten rats survived the surgery and testing period with no or mild temporary neurological compromise. The study revealed that carotid catheters filled with heparinized glycerol ensure better catheter patency rates and blood pressure transduction. There was no significant drift in the device's pressure sensitivity during the experiment. To our knowledge, this is the first experimental study to show considerable animal survival after lower medullary implantation. Combining the ability to measure and monitor invasive blood pressure with a closed-loop brain pulse generator in a single device could be of potential value in future hemodynamic animal research.
Keywords: caudal ventrolateral medulla; closed-loop stimulation; deep brain stimulation; hypertension; rat model; telemetry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources

