Identification of a Ubiquinone-Ubiquinol Quinhydrone Complex in Bacterial Photosynthetic Membranes and Isolated Reaction Centers by Time-Resolved Infrared Spectroscopy
- PMID: 36982307
- PMCID: PMC10049466
- DOI: 10.3390/ijms24065233
Identification of a Ubiquinone-Ubiquinol Quinhydrone Complex in Bacterial Photosynthetic Membranes and Isolated Reaction Centers by Time-Resolved Infrared Spectroscopy
Abstract
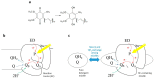
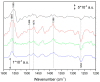
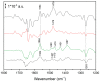
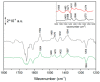
Ubiquinone redox chemistry is of fundamental importance in biochemistry, notably in bioenergetics. The bi-electronic reduction of ubiquinone to ubiquinol has been widely studied, including by Fourier transform infrared (FTIR) difference spectroscopy, in several systems. In this paper, we have recorded static and time-resolved FTIR difference spectra reflecting light-induced ubiquinone reduction to ubiquinol in bacterial photosynthetic membranes and in detergent-isolated photosynthetic bacterial reaction centers. We found compelling evidence that in both systems under strong light illumination-and also in detergent-isolated reaction centers after two saturating flashes-a ubiquinone-ubiquinol charge-transfer quinhydrone complex, characterized by a characteristic band at ~1565 cm-1, can be formed. Quantum chemistry calculations confirmed that such a band is due to formation of a quinhydrone complex. We propose that the formation of such a complex takes place when Q and QH2 are forced, by spatial constraints, to share a common limited space as, for instance, in detergent micelles, or when an incoming quinone from the pool meets, in the channel for quinone/quinol exchange at the QB site, a quinol coming out. This latter situation can take place both in isolated and membrane bound reaction centers Possible consequences of the formation of this charge-transfer complex under physiological conditions are discussed.
Keywords: Fourier Transform Infrared (FTIR) difference spectroscopy; Rhodobacter (Rb.) sphaeroides; bacterial reaction center (RC); charge-transfer complex; chromatophores; quinhydrone; rapid-scan FTIR; ubiquinol; ubiquinone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Trumpower B.L. Function of Quinones in Energy Conserving Systems. Academic Press; New York, NY, USA: 1982.
-
- Feher G., Allen J.P., Okamura M.Y., Rees D.C. Structure and function of bacterial photosynthetic reaction centres. Nature. 1989;339:111–116. doi: 10.1038/339111a0. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

