Regulation of Cell Plasticity by Bromodomain and Extraterminal Domain (BET) Proteins: A New Perspective in Glioblastoma Therapy
- PMID: 36982740
- PMCID: PMC10055343
- DOI: 10.3390/ijms24065665
Regulation of Cell Plasticity by Bromodomain and Extraterminal Domain (BET) Proteins: A New Perspective in Glioblastoma Therapy
Abstract
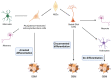
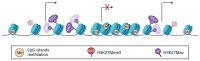
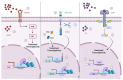
BET proteins are a family of multifunctional epigenetic readers, mainly involved in transcriptional regulation through chromatin modelling. Transcriptome handling ability of BET proteins suggests a key role in the modulation of cell plasticity, both in fate decision and in lineage commitment during embryonic development and in pathogenic conditions, including cancerogenesis. Glioblastoma is the most aggressive form of glioma, characterized by a very poor prognosis despite the application of a multimodal therapy. Recently, new insights are emerging about the glioblastoma cellular origin, leading to the hypothesis that several putative mechanisms occur during gliomagenesis. Interestingly, epigenome dysregulation associated with loss of cellular identity and functions are emerging as crucial features of glioblastoma pathogenesis. Therefore, the emerging roles of BET protein in glioblastoma onco-biology and the compelling demand for more effective therapeutic strategies suggest that BET family members could be promising targets for translational breakthroughs in glioblastoma treatment. Primarily, "Reprogramming Therapy", which is aimed at reverting the malignant phenotype, is now considered a promising strategy for GBM therapy.
Keywords: BET proteins; cell plasticity; differentiation; epigenome; glioblastoma; reprogramming therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Ostrom Q.T., Gittleman H., Liao P., Vecchione-Koval T., Wolinsky Y., Kruchko C., Barnholtz-Sloan J.S. CBTRUS Statistical Report: Primary brain and other central nervous system tumors diagnosed in the United States in 2010–2014. Neuro. Oncol. 2017;19:v1–v88. doi: 10.1093/neuonc/nox158. - DOI - PMC - PubMed
-
- Stupp R., Hegi M.E., Mason W.P., van den Bent M.J., Taphoorn M.J., Janzer R.C., Ludwin S.K., Allgeier A., Fisher B., Belanger K., et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. - DOI - PubMed
-
- Janjua T.I., Rewatkar P., Ahmed-Cox A., Saeed I., Mansfeld F.M., Kulshreshtha R., Kumeria T., Ziegler D.S., Kavallaris M., Mazzieri R., et al. Frontiers in the treatment of glioblastoma: Past, present and emerging. Adv. Drug Deliv. Rev. 2021;171:108–138. doi: 10.1016/j.addr.2021.01.012. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

