Combined Inhibition of Hedgehog and HDAC6: In Vitro and In Vivo Studies Reveal a New Role for Lysosomal Stress in Reducing Glioblastoma Cell Viability
- PMID: 36982845
- PMCID: PMC10051748
- DOI: 10.3390/ijms24065771
Combined Inhibition of Hedgehog and HDAC6: In Vitro and In Vivo Studies Reveal a New Role for Lysosomal Stress in Reducing Glioblastoma Cell Viability
Abstract
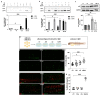
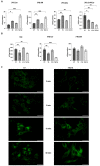
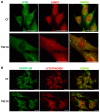
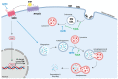
Glioblastoma multiforme (GBM) is the most common and malignant brain tumor in adults. The invasiveness and the rapid progression that characterize GBM negatively impact patients' survival. Temozolomide (TMZ) is currently considered the first-choice chemotherapeutic agent. Unfortunately, over 50% of patients with GBM do not respond to TMZ treatment, and the mutation-prone nature of GBM enables the development of resistance mechanisms. Therefore, efforts have been devoted to the dissection of aberrant pathways involved in GBM insurgence and resistance in order to identify new therapeutic targets. Among them, sphingolipid signaling, Hedgehog (Hh) pathway, and the histone deacetylase 6 (HDAC6) activity are frequently dysregulated and may represent key targets to counteract GBM progression. Given the positive correlation between Hh/HDAC6/sphingolipid metabolism in GBM, we decided to perform a dual pharmacological inhibition of Hh and HDAC6 through cyclopamine and tubastatin A, respectively, in a human GMB cell line and zebrafish embryos. The combined administration of these compounds elicited a more significant reduction of GMB cell viability than did single treatments in vitro and in cells orthotopically transplanted in the zebrafish hindbrain ventricle. We demonstrated, for the first time, that the inhibition of these pathways induces lysosomal stress which results in an impaired fusion of lysosomes with autophagosomes and a block of sphingolipid degradation in GBM cell lines. This condition, which we also recapitulated in zebrafish embryos, suggests an impairment of lysosome-dependent processes involving autophagy and sphingolipid homeostasis and might be instrumental in the reduction of GBM progression.
Keywords: HDAC6; Hh; autophagy; combination treatment; glioblastoma; lysosomal stress; sphingolipids; zebrafish.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

