Platinum-Nucleos(t)ide Compounds as Possible Antimetabolites for Antitumor/Antiviral Therapy: Properties and Perspectives
- PMID: 36986802
- PMCID: PMC10058173
- DOI: 10.3390/pharmaceutics15030941
Platinum-Nucleos(t)ide Compounds as Possible Antimetabolites for Antitumor/Antiviral Therapy: Properties and Perspectives
Abstract
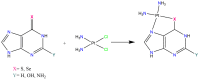
Nucleoside analogues (NAs) are a family of compounds which include a variety of purine and pyrimidine derivatives, widely used as anticancer and antiviral agents. For their ability to compete with physiological nucleosides, NAs act as antimetabolites exerting their activity by interfering with the synthesis of nucleic acids. Much progress in the comprehension of their molecular mechanisms has been made, including providing new strategies for potentiating anticancer/antiviral activity. Among these strategies, new platinum-NAs showing a good potential to improve the therapeutic indices of NAs have been synthesized and studied. This short review aims to describe the properties and future perspectives of platinum-NAs, proposing these complexes as a new class of antimetabolites.
Keywords: antimetabolites; antitumor drugs; antiviral drugs; coordination compounds; nucleoside analogues; platinum compounds.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



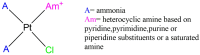




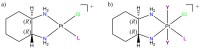



References
-
- Mahmoud S., Hasabelnaby S., Hammad S., Sakr T. Antiviral Nucleoside and Nucleotide Analogs: A Review. J. Adv. Pharm. Res. 2018;2:73–88. doi: 10.21608/aprh.2018.5829. - DOI
Publication types
LinkOut - more resources
Full Text Sources

