Vaccination against Varicella Zoster Virus Infection in Less Developed Regions of Guangdong, China: A Cross-Sectional Serosurveillance Study
- PMID: 36992078
- PMCID: PMC10056728
- DOI: 10.3390/vaccines11030494
Vaccination against Varicella Zoster Virus Infection in Less Developed Regions of Guangdong, China: A Cross-Sectional Serosurveillance Study
Abstract
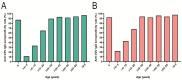
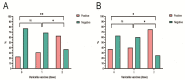
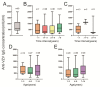
Vaccination is the key to prevent varicella zoster virus (VZV) infection in children. Voluntary and self-funded strategies have led to variable vaccination rates against VZV in China. For low-income populations, in particular, the effects of VZV vaccination have been insufficiently estimated. Community-based serosurveillance was conducted in two less developed regions, Zhanjiang and Heyuan, of Guangdong, China. Anti-VZV IgG antibodies in serum were detected by ELISA. The vaccination data were derived from the Guangdong Immune Planning Information System. A total of 4221 participants were involved, of which 3377 were from three counties of Zhanjiang and the other 844 were from one county of Heyuan, Guangdong, China. The total VZV IgG seropositivity rate in vaccinated individuals was 34.30% and 42.76%, while it was 89.61% and 91.62% in non-vaccinated populations of Zhanjiang and Heyuan, respectively. The seropositivity rate increased gradually with age, reaching ~90% in the >20- to 30-year-old group. The VarV vaccination rates of children aged 1-14 years were 60.47% for one dose and 6.20% for two doses in Zhanjiang, and 52.24% for one dose and 4.48% for two doses in Heyuan. Compared with the non-vaccinated group (31.19%) and one-dose group (35.47%), the positivity rate of anti-VZV IgG antibodies was significantly higher in the two-dose group (67.86%). Before the VarV policy was reformed, the anti-VZV IgG positivity rate was 27.85% in the one-dose-vaccinated participants, which increased to 30.43% after October 2017. The high seroprevalence in participants was due to infection of VZV in Zhanjiang and Heyuan, not vaccination against VZV. Children aged 0-5 years are still vulnerable to varicella, so a two-dose vaccination program should be implemented to prevent onward transmission of VZV.
Keywords: Guangdong; anti-VZV IgG; seroprevalence; undeveloped; vaccination; varicella.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Seroepidemiology of varicella in Hangzhou, China in the vaccine era.Hum Vaccin Immunother. 2018;14(10):2464-2471. doi: 10.1080/21645515.2018.1477909. Epub 2018 Jul 30. Hum Vaccin Immunother. 2018. PMID: 30019992 Free PMC article.
-
Seroprevalence and molecular characteristics of varicella-zoster virus infection in Chinese children.BMC Infect Dis. 2019 Jul 19;19(1):643. doi: 10.1186/s12879-019-4233-7. BMC Infect Dis. 2019. PMID: 31324226 Free PMC article.
-
Nosocomial transmission of chickenpox and varicella zoster virus seroprevalence rate amongst healthcare workers in a teaching hospital in China.BMC Infect Dis. 2019 Jul 5;19(1):582. doi: 10.1186/s12879-019-4222-x. BMC Infect Dis. 2019. PMID: 31277589 Free PMC article.
-
Vaccines for preventing herpes zoster in older adults.Cochrane Database Syst Rev. 2019 Nov 7;2019(11):CD008858. doi: 10.1002/14651858.CD008858.pub4. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2023 Oct 2;10:CD008858. doi: 10.1002/14651858.CD008858.pub5. PMID: 31696946 Free PMC article. Updated.
-
Varicella vaccination in Japan: necessity of implementing a routine vaccination program.J Infect Chemother. 2013 Apr;19(2):188-95. doi: 10.1007/s10156-013-0577-x. Epub 2013 Mar 13. J Infect Chemother. 2013. PMID: 23483311 Review.
Cited by
-
Optimizing Varicella Vaccination Strategy: A Study on Age and Dose Impacts on Antibody Levels.Vaccines (Basel). 2024 Dec 30;13(1):23. doi: 10.3390/vaccines13010023. Vaccines (Basel). 2024. PMID: 39852802 Free PMC article.
-
Immunogenicity and Safety of a Newly Developed Live Attenuated Varicella Vaccine in Healthy Children: A Multi-National, Randomized, Double-Blinded, Active-Controlled, Phase 3 Study.Vaccines (Basel). 2023 Aug 24;11(9):1416. doi: 10.3390/vaccines11091416. Vaccines (Basel). 2023. PMID: 37766093 Free PMC article.
References
-
- Sadaoka T., Mori Y. Vaccine Development for Varicella-Zoster Virus. Adv. Exp. Med. Biol. 2018;1045:123–142. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

