Epstein-Barr Virus History and Pathogenesis
- PMID: 36992423
- PMCID: PMC10056551
- DOI: 10.3390/v15030714
Epstein-Barr Virus History and Pathogenesis
Abstract
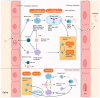
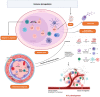
Epstein-Barr virus (EBV) is the first identified human oncogenic virus that can establish asymptomatic life-long persistence. It is associated with a large spectrum of diseases, including benign diseases, a number of lymphoid malignancies, and epithelial cancers. EBV can also transform quiescent B lymphocytes into lymphoblastoid cell lines (LCLs) in vitro. Although EBV molecular biology and EBV-related diseases have been continuously investigated for nearly 60 years, the mechanism of viral-mediated transformation, as well as the precise role of EBV in promoting these diseases, remain a major challenge yet to be completely explored. This review will highlight the history of EBV and current advances in EBV-associated diseases, focusing on how this virus provides a paradigm for exploiting the many insights identified through interplay between EBV and its host during oncogenesis, and other related non-malignant disorders.
Keywords: Epstein-Barr virus; cancer induction; epithelial carcinoma; lymphoma; pathogenesis.
Conflict of interest statement
The authors declare no conflict of interest. The funder had no role in the design of the study, in the writing of the manuscript, or in the decision to publish the results.
Figures
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

