This is a preprint.
Bacterial contact induces polar plug disintegration to mediate whipworm egg hatching
- PMID: 36993552
- PMCID: PMC10054943
- DOI: 10.1101/2023.03.13.532458
Bacterial contact induces polar plug disintegration to mediate whipworm egg hatching
Update in
-
Bacterial contact induces polar plug disintegration to mediate whipworm egg hatching.PLoS Pathog. 2023 Sep 22;19(9):e1011647. doi: 10.1371/journal.ppat.1011647. eCollection 2023 Sep. PLoS Pathog. 2023. PMID: 37738244 Free PMC article.
Abstract
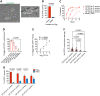
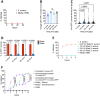
The bacterial microbiota promotes the life cycle of the intestine-dwelling whipworm Trichuris by mediating hatching of parasite eggs ingested by the mammalian host. Despite the enormous disease burden associated with Trichuris colonization, the mechanisms underlying this transkingdom interaction have been obscure. Here, we used a multiscale microscopy approach to define the structural events associated with bacteria-mediated hatching of eggs for the murine model parasite Trichuris muris . Through the combination of scanning electron microscopy (SEM) and serial block face SEM (SBFSEM), we visualized the outer surface morphology of the shell and generated 3D structures of the egg and larva during the hatching process. These images revealed that exposure to hatching-inducing bacteria catalyzed asymmetric degradation of the polar plugs prior to exit by the larva. Although unrelated bacteria induced similar loss of electron density and dissolution of the structural integrity of the plugs, egg hatching was most efficient in the presence of bacteria that bound poles with high density such as Staphylococcus aureus . Consistent with the ability of taxonomically distant bacteria to induce hatching, additional results suggest chitinase released from larva within the eggs degrade the plugs from the inside instead of enzymes produced by bacteria in the external environment. These findings define at ultrastructure resolution the evolutionary adaptation of a parasite for the microbe-rich environment of the mammalian gut.
Figures





References
-
- Soil-transmitted helminth infections 2020 [updated 2nd March, 2020. Available from: https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helmin....
-
- Panesar TS, Croll NA. The location of parasites within their hosts: Site selection by Trichuris muris in the laboratory mouse. International Journal for Parasitology. 1980;10(4):261–73. - PubMed
-
- Preston CM, Jenkins T. Trichuris muris: Structure and formation of the egg polar plugs. Zeitschrift für Parasitenkunde. 1985;71(3):373–81. - PubMed
-
- Brooker SJ, Mwandawiro CS, Halliday KE, Njenga SM, McHaro C, Gichuki PM, et al. Interrupting transmission of soil-transmitted helminths: a study protocol for cluster randomised trials evaluating alternative treatment strategies and delivery systems in Kenya. BMJ Open. 2015;5(10):e008950. - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
