Characterization of "Off-Target" Immune Modulation Induced by Live Attenuated Yellow Fever Vaccine
- PMID: 36994927
- PMCID: PMC10938199
- DOI: 10.1093/infdis/jiad086
Characterization of "Off-Target" Immune Modulation Induced by Live Attenuated Yellow Fever Vaccine
Abstract
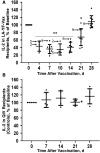
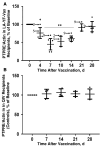
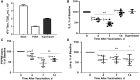
Background: Live attenuated vaccines alter immune functions and are associated with beneficial outcomes. We previously demonstrated that live attenuated yellow fever virus (YFV) vaccine (LA-YF-Vax) dampens T-cell receptor (TCR) signaling in vitro via an RNA-based mechanism. We examined study participants before and after LA-YF-Vax to assess TCR-mediated functions in vivo.
Methods: Serum samples and peripheral blood mononuclear cells (PBMCs) were obtained before and after LA-YF-Vax (with or without additional vaccines) or quadrivalent influenza vaccine. TCR-mediated activation was determined by interleukin 2 release or phosphorylation of the lymphocyte-specific Src kinase. TCR-regulating phosphatase (protein tyrosine phosphatase receptor type E [PTPRE]) expression was also measured.
Results: Compared with prevaccination findings, LA-YF-Vax recipient PBMCs demonstrated transient reduction in interleukin 2 release after TCR stimulation and PTPRE levels, unlike in control participants who received quadrivalent influenza vaccine. YFV was detected in 8 of 14 participants after LA-YF-Vax. After incubation of healthy donor PBMCs in serum-derived extracellular vesicles prepared from LA-YF-Vax recipients, TCR signaling and PTPRE levels were reduced after vaccination, even in participants without detectable YFV RNA.
Conclusions: LA-YF-Vax reduces TCR functions and PTPRE levels after vaccination. Extracellular vesicles from serum recapitulated this effect in healthy cells. This likely contributes to the reduced immunogenicity for heterologous vaccines after LA-YF-Vax administration. Identification of specific immune mechanisms related to vaccines should contribute to understanding of the "off-target," beneficial effects of live vaccines.
Keywords: Off-target vaccine effects; PTPRE; T lymphocyte; T-cell receptor; live attenuated yellow fever vaccine; vaccine; yellow fever virus.
Published by Oxford University Press on behalf of Infectious Diseases Society of America 2023.
Conflict of interest statement
Potential conflicts of interest . All authors: No reported conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Figures
Similar articles
-
Safety of live vaccinations on immunosuppressive therapy in patients with immune-mediated inflammatory diseases, solid organ transplantation or after bone-marrow transplantation - A systematic review of randomized trials, observational studies and case reports.Vaccine. 2017 Mar 1;35(9):1216-1226. doi: 10.1016/j.vaccine.2017.01.048. Epub 2017 Feb 3. Vaccine. 2017. PMID: 28162821
-
Yellow fever vaccine for patients with HIV infection.Cochrane Database Syst Rev. 2014 Jan 23;2014(1):CD010929. doi: 10.1002/14651858.CD010929.pub2. Cochrane Database Syst Rev. 2014. PMID: 24453061 Free PMC article.
-
Yellow Fever Virus, but Not Zika Virus or Dengue Virus, Inhibits T-Cell Receptor-Mediated T-Cell Function by an RNA-Based Mechanism.J Infect Dis. 2017 Nov 27;216(9):1164-1175. doi: 10.1093/infdis/jix462. J Infect Dis. 2017. PMID: 28968905 Free PMC article.
-
Transmission of yellow fever vaccine virus from breast feeding mothers to their infants: reporting of yellow fever virus (YFV) RNA detection in milk specimens.F1000Res. 2024 Feb 29;11:76. doi: 10.12688/f1000research.74576.4. eCollection 2022. F1000Res. 2024. PMID: 38106444 Free PMC article.
-
Two mutations in NS2B are responsible for attenuation of the yellow fever virus (YFV) vaccine strain 17D.PLoS Pathog. 2025 Jul 31;21(7):e1013373. doi: 10.1371/journal.ppat.1013373. eCollection 2025 Jul. PLoS Pathog. 2025. PMID: 40743291 Free PMC article.
Cited by
-
Antiretroviral Therapy Suppresses RNA N6-Methyladenosine Modification in Peripheral Blood Mononuclear Cells from HIV-1-Infected Individuals.AIDS Res Hum Retroviruses. 2024 Sep;40(9):511-520. doi: 10.1089/AID.2024.0003. Epub 2024 May 23. AIDS Res Hum Retroviruses. 2024. PMID: 38753726
References
-
- Jensen KJ, Benn CS, van Crevel R. Unravelling the nature of non-specific effects of vaccines-a challenge for innate immunologists. Semin Immunol 2016; 28:377–83. - PubMed
-
- Benn CS, Netea MG, Selin LK, Aaby P. A small jab—a big effect: nonspecific immunomodulation by vaccines. Trends Immunol 2013; 34:431–9. - PubMed
-
- Biering-Sørensen S, Aaby P, Napirna BM, et al. Small randomized trial among low-birth-weight children receiving bacillus Calmette–Guerin vaccination at first health center contact. Pediatr Infect Dis J 2012; 31:306–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

