Neuro-oncological augmented reality planning for intracranial tumor resection
- PMID: 36998774
- PMCID: PMC10043492
- DOI: 10.3389/fneur.2023.1104571
Neuro-oncological augmented reality planning for intracranial tumor resection
Abstract
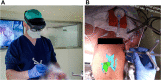
Background: Before starting surgery for the resection of an intracranial tumor, its outlines are typically marked on the skin of the patient. This allows for the planning of the optimal skin incision, craniotomy, and angle of approach. Conventionally, the surgeon determines tumor borders using neuronavigation with a tracked pointer. However, interpretation errors can lead to important deviations, especially for deep-seated tumors, potentially resulting in a suboptimal approach with incomplete exposure. Augmented reality (AR) allows displaying of the tumor and critical structures directly on the patient, which can simplify and improve surgical preparation.
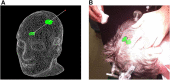
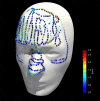
Methods: We developed an AR-based workflow for intracranial tumor resection planning deployed on the Microsoft HoloLens II, which exploits the built-in infrared-camera for tracking the patient. We initially performed a phantom study to assess the accuracy of the registration and tracking. Following this, we evaluated the AR-based planning step in a prospective clinical study for patients undergoing resection of a brain tumor. This planning step was performed by 12 surgeons and trainees with varying degrees of experience. After patient registration, tumor outlines were marked on the patient's skin by different investigators, consecutively using a conventional neuronavigation system and an AR-based system. Their performance in both registration and delineation was measured in terms of accuracy and duration and compared.
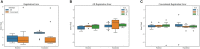
Results: During phantom testing, registration errors remained below 2.0 mm and 2.0° for both AR-based navigation and conventional neuronavigation, with no significant difference between both systems. In the prospective clinical trial, 20 patients underwent tumor resection planning. Registration accuracy was independent of user experience for both AR-based navigation and the commercial neuronavigation system. AR-guided tumor delineation was deemed superior in 65% of cases, equally good in 30% of cases, and inferior in 5% of cases when compared to the conventional navigation system. The overall planning time (AR = 119 ± 44 s, conventional = 187 ± 56 s) was significantly reduced through the adoption of the AR workflow (p < 0.001), with an average time reduction of 39%.
Conclusion: By providing a more intuitive visualization of relevant data to the surgeon, AR navigation provides an accurate method for tumor resection planning that is quicker and more intuitive than conventional neuronavigation. Further research should focus on intraoperative implementations.
Keywords: augmented reality; brain tumor; computer-assisted surgery; intracranial tumor; neuronavigation; preoperative preparation; resection planning.
Copyright © 2023 Van Gestel, Frantz, Buyck, Geens, Neuville, Bruneau, Jansen, Scheerlinck, Vandemeulebroucke and Duerinck.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures









References
-
- Kunz C, Maurer P, Kees F, Henrich P, Marzi C, Hlaváč M, et al. . Infrared marker tracking with the HoloLens for neurosurgical interventions. Curr Dir Biomed Eng. (2020) 6:20200027. 10.1515/cdbme-2020-0027 - DOI
LinkOut - more resources
Full Text Sources
Research Materials

