Attack phenotypes and disease course in pediatric MOGAD
- PMID: 37000895
- PMCID: PMC10187731
- DOI: 10.1002/acn3.51759
Attack phenotypes and disease course in pediatric MOGAD
Abstract
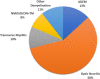
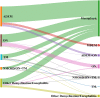
Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) is an autoimmune demyelinating condition that affects children differently than adults. We performed a literature review to assess the presentation and clinical course of pediatric MOGAD. The most common initial phenotype is acute disseminated encephalomyelitis, especially among children younger than five years, followed by optic neuritis (ON) and/or transverse myelitis. Approximately one-quarter of children with MOGAD have at least one relapse that typically occurs within three years of disease onset and often includes ON, even if ON was not present at onset. Clinical risk factors for a relapsing course have not been elucidated.
© 2023 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
This study was funded by UCB Pharma. Medical writing support was provided by Epi Excellence LLC (Garnet Valley, PA, USA) and funded by UCB Pharma, in accordance with Good Publication Practice (GPP3) guidelines (
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
