Multicomponent synthesis of chromophores - The one-pot approach to functional π-systems
- PMID: 37007054
- PMCID: PMC10065161
- DOI: 10.3389/fchem.2023.1124209
Multicomponent synthesis of chromophores - The one-pot approach to functional π-systems
Abstract
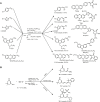
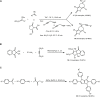
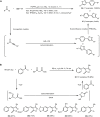
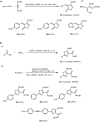
Multicomponent reactions, conducted in a domino, sequential or consecutive fashion, have not only considerably enhanced synthetic efficiency as one-pot methodology, but they have also become an enabling tool for interdisciplinary research. The highly diversity-oriented nature of the synthetic concept allows accessing huge structural and functional space. Already some decades ago this has been recognized for life sciences, in particular, lead finding and exploration in pharma and agricultural chemistry. The quest for novel functional materials has also opened the field for diversity-oriented syntheses of functional π-systems, i.e. dyes for photonic and electronic applications based on their electronic properties. This review summarizes recent developments in MCR syntheses of functional chromophores highlighting syntheses following either the framework forming scaffold approach by establishing connectivity between chromophores or the chromogenic chromophore approach by de novo formation of chromophore of interest. Both approaches warrant rapid access to molecular functional π-systems, i.e. chromophores, fluorophores, and electrophores for various applications.
Keywords: absorption; chromophores; diversity-oriented synthesis; donor-acceptor systems; fluorescence; multicomponent reactions; one-pot synthesis.
Copyright © 2023 Brandner and Müller.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







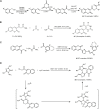

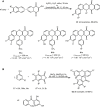
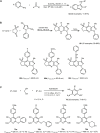

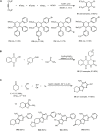

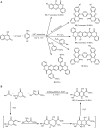
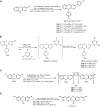
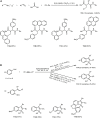
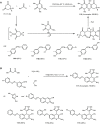
Similar articles
-
Multicomponent syntheses of functional chromophores.Chem Soc Rev. 2016 May 21;45(10):2825-46. doi: 10.1039/c5cs00805k. Epub 2016 Feb 22. Chem Soc Rev. 2016. PMID: 26898222
-
Three-Component Activation/Alkynylation/Cyclocondensation (AACC) Synthesis of Enhanced Emission Solvatochromic 3-Ethynylquinoxalines.Chemistry. 2018 Jun 7;24(32):8114-8125. doi: 10.1002/chem.201800079. Epub 2018 Mar 30. Chemistry. 2018. PMID: 29425410
-
Multicomponent syntheses of pyrazoles via (3 + 2)-cyclocondensation and (3 + 2)-cycloaddition key steps.Beilstein J Org Chem. 2024 Aug 16;20:2024-2077. doi: 10.3762/bjoc.20.178. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 39161713 Free PMC article. Review.
-
Multicomponent Hetero-[4 + 2] Cycloaddition/Allylboration Reaction: From Natural Product Synthesis to Drug Discovery.Acc Chem Res. 2016 Nov 15;49(11):2489-2500. doi: 10.1021/acs.accounts.6b00403. Epub 2016 Oct 18. Acc Chem Res. 2016. PMID: 27753496
-
Functional chromophores synthesized via multicomponent Reactions: A review on their use as cell-imaging probes.Methods. 2023 Dec;220:142-157. doi: 10.1016/j.ymeth.2023.11.001. Epub 2023 Nov 6. Methods. 2023. PMID: 37939912 Review.
Cited by
-
Modular Design and Scaffold-Synthesis of Multi-Functional Fluorophores for Targeted Cellular Imaging and Pyroptosis.Angew Chem Int Ed Engl. 2025 Jan 21;64(4):e202415627. doi: 10.1002/anie.202415627. Epub 2024 Nov 18. Angew Chem Int Ed Engl. 2025. PMID: 39555698 Free PMC article.
-
Lab-on-a-molecule and multi-analyte sensing.Front Chem. 2024 Apr 18;12:1393308. doi: 10.3389/fchem.2024.1393308. eCollection 2024. Front Chem. 2024. PMID: 38698939 Free PMC article. Review.
-
Computer-Generated, Mechanistic Networks Assist in Assigning the Outcomes of Complex Multicomponent Reactions.J Am Chem Soc. 2025 May 7;147(18):15636-15644. doi: 10.1021/jacs.5c02846. Epub 2025 Apr 28. J Am Chem Soc. 2025. PMID: 40293375 Free PMC article.
-
Alkynylated and triazole-linked aroyl-S,N-ketene acetals: one-pot synthesis of solid-state emissive dyes with aggregation-induced enhanced emission characteristics.Sci Rep. 2023 Sep 1;13(1):14399. doi: 10.1038/s41598-023-41146-w. Sci Rep. 2023. PMID: 37658089 Free PMC article.
-
Facile One-Pot Fischer-Suzuki-Knoevenagel Microwave-Assisted Synthesis of Fluorescent 5-Aryl-2-Styryl-3H-Indoles.Molecules. 2025 Jun 7;30(12):2503. doi: 10.3390/molecules30122503. Molecules. 2025. PMID: 40572469 Free PMC article.
References
-
- Abaee M. S., Hadizadeh A., Mojtahedi M. M., Halvagar M. R. (2017). Exploring the scope of the Gewald reaction: Expansion to a four-component process. Tetrahedron Lett. 58, 1408–1412. 10.1016/j.tetlet.2017.02.071 - DOI
-
- Abdel-Mottaleb M. S. A., Antonious M. S., Ali M. M. A., Ismail L. F. M., El-Sayed B. A., Sherief A. M. K. (1992). Photophysics and dynamics of coumarin laser dyes and their analytical implications. Proc. Indian Acad. Sci. 104, 185. 10.1007/BF02863363 - DOI
-
- Abu-Reziq R., Wang D., Post M., Alper H. (2008). Separable catalysts in one-pot syntheses for greener chemistry. Chem. Mat. 20, 2544–2550. 10.1021/cm703208w - DOI
-
- Affeldt R. F., de Amorim Borges A. C., Russowsky D., Severo Rodembusch F. (2014). Synthesis and fluorescence properties of benzoxazole-1,4-dihydropyridine dyads achieved by a multicomponent reaction. New J. Chem. 38, 4607–4614. 10.1039/C4NJ00777H - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

