Quantitative Evaluation of a Multimodal Aptamer-Targeted Long-Circulating Polymer for Tumor Targeting
- PMID: 37008162
- PMCID: PMC10061651
- DOI: 10.1021/acsomega.2c07762
Quantitative Evaluation of a Multimodal Aptamer-Targeted Long-Circulating Polymer for Tumor Targeting
Abstract
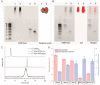
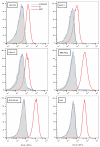
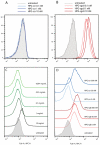
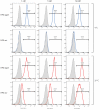
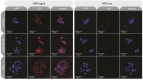
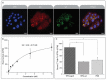
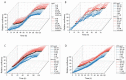
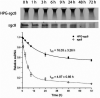
Aptamers are promising targeting agents for imaging and therapy of numerous diseases, including cancer. However, a significant shortcoming of aptamers is their poor stability and fast excretion, limiting their application in vivo. Common strategies to overcome these challenges is to chemically modify aptamers in order to increase their stability and/or to apply formulation technologies such as conjugating them to polymers or nanocarriers in order to increase their circulation half-life. This is expected to result in improved cellular uptake or retention to passively targeted nanomedicines. Herein, we report a modular conjugation strategy based on click chemistry between functionalized tetrazines and trans-cyclooctene (TCO), for the modification of high molecular weight hyperbranched polyglycerol (HPG) with sgc8 aptamer, fluorescent dyes, and 111In. Our data indicate strong affinity of sgc8 against a range of solid tumor-derived cell lines that have previously not been tested with this aptamer. Nevertheless, nonspecific uptake of scrambled ssDNA-functionalized HPG in cells highlights inherent challenges of aptamer-targeted probes that remain to be solved for clinical translation. We validate HPG-sgc8 as a nontoxic nanoprobe with high affinity against MDA-MB-468 breast and A431 lung cancer cells and show significantly increased plasma stability compared to free sgc8. In vivo quantitative SPECT/CT imaging indicates EPR-mediated tumor uptake of HPG-sgc8 and nontargeted or scrambled ssDNA-conjugated HPG but no statistically significant difference between these formulations in terms of total tumor uptake or retention. Our study emphasizes the need for stringent controls and quantification in the evaluation of aptamer-targeted probes. For this purpose, our versatile synthesis strategy provides a simple approach for the design and evaluation of long-circulating aptamer-conjugated nanoformulations.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

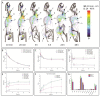
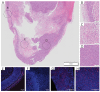
References
-
- Chapman S.; Dobrovolskaia M.; Farahani K.; Goodwin A.; Joshi A.; Lee H.; Meade T.; Pomper M.; Ptak K.; Rao J.; Singh R.; Sridhar S.; Stern S.; Wang A.; Weaver J. B.; Woloschak G.; Yang L. Nanoparticles for cancer imaging: The good, the bad, and the promise. Nano Today 2013, 8 (5), 454–460. 10.1016/j.nantod.2013.06.001. - DOI - PMC - PubMed
-
- Kainthan R. K.; Muliawan E. B.; Hatzikiriakos S. G.; Brooks D. E. Synthesis, Characterization, and Viscoelastic Properties of High Molecular Weight Hyperbranched Polyglycerols. Macromolecules 2006, 39 (22), 7708–7717. 10.1021/ma0613483. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

