Effects of intrauterine growth restriction on embryonic hippocampal dentate gyrus neurogenesis and postnatal critical period of synaptic plasticity that govern learning and memory function
- PMID: 37008232
- PMCID: PMC10064986
- DOI: 10.3389/fnins.2023.1092357
Effects of intrauterine growth restriction on embryonic hippocampal dentate gyrus neurogenesis and postnatal critical period of synaptic plasticity that govern learning and memory function
Abstract
Intrauterine growth restriction (IUGR) complicates up to 10% of human pregnancies and is the second leading cause of perinatal morbidity and mortality after prematurity. The most common etiology of IUGR in developed countries is uteroplacental insufficiency (UPI). For survivors of IUGR pregnancies, long-term studies consistently show a fivefold increased risk for impaired cognition including learning and memory deficits. Among these, only a few human studies have highlighted sex differences with males and females having differing susceptibilities to different impairments. Moreover, it is well established from brain magnetic resonance imaging that IUGR affects both white and gray matter. The hippocampus, composed of the dentate gyrus (DG) and cornu ammonis (CA) subregions, is an important gray matter structure critical to learning and memory, and is particularly vulnerable to the chronic hypoxic-ischemic effects of UPI. Decreased hippocampal volume is a strong predictor for learning and memory deficits. Decreased neuron number and attenuated dendritic and axonal morphologies in both the DG and CA are additionally seen in animal models. What is largely unexplored is the prenatal changes that predispose an IUGR offspring to postnatal learning and memory deficits. This lack of knowledge will continue to hinder the design of future therapy to improve learning and memory. In this review, we will first present the clinical susceptibilities and human epidemiology data regarding the neurological sequelae after IUGR. We will follow with data generated using our laboratory's mouse model of IUGR, that mimics the human IUGR phenotype, to dissect at the cellular and molecular alterations in embryonic hippocampal DG neurogenesis. We will lastly present a newer topic of postnatal neuron development, namely the critical period of synaptic plasticity that is crucial in achieving an excitatory/inhibitory balance in the developing brain. To our knowledge, these findings are the first to describe the prenatal changes that lead to an alteration in postnatal hippocampal excitatory/inhibitory imbalance, a mechanism that is now recognized to be a cause of neurocognitive/neuropsychiatric disorders in at-risk individuals. Studies are ongoing in our laboratory to elucidate additional mechanisms that underlie IUGR-induced learning and memory impairment and to design therapy aimed at ameliorating such impairment.
Keywords: Wnt signaling; critical period of synaptic plasticity; embryonic neurogenesis; fetal growth restriction; hippocampal dentate gyrus; hypertensive disease of pregnancy; intrauterine growth restriction; learning and memory.
Copyright © 2023 Fung.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
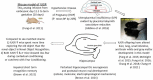


References
-
- Aceti M., Creson T. K., Vaissiere T., Rojas C., Huang W. C., Wang Y. X., et al. (2015). Syngap1 haploinsufficiency damages a postnatal critical period of pyramidal cell structural maturation linked to cortical circuit assembly. Biol. Psychiatry 77 805–815. 10.1016/j.biopsych.2014.08.001 - DOI - PMC - PubMed
-
- Batalle D., Eixarch E., Figueras F., Munoz-Moreno E., Bargallo N., Illa M., et al. (2012). Altered small-world topology of structural brain networks in infants with intrauterine growth restriction and its association with later neurodevelopmental outcome. Neuroimage 60 1352–1366. 10.1016/j.neuroimage.2012.01.059 - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

