Pharmacological Evaluation of the Anesthetic and Analgesic Potential of Injection Harsha 22: A Novel Polyherbal Local Anesthetic Formulation Intended for Parenteral Administration in Wistar Albino Rats
- PMID: 37008368
- PMCID: PMC10065419
- DOI: 10.2147/JEP.S402277
Pharmacological Evaluation of the Anesthetic and Analgesic Potential of Injection Harsha 22: A Novel Polyherbal Local Anesthetic Formulation Intended for Parenteral Administration in Wistar Albino Rats
Abstract
Background: Local anaesthetics are medications that cause numbness that can be reversed by applying them topically. Local anaesthetics are clinically used to control pain during minor surgeries or to treat other acute and chronic pain. The present investigation intended to investigate the anesthetic as well as analgesic potential of Injection Harsha 22, a novel polyherbal formulation in Wistar albino rats.
Methods: The anesthetic potential of Injection Harsha 22 was evaluated by a heat tail-flick latency (TFL) test, whereas the analgesic effect was elevated by electrical stimulation testing. Here, lignocaine (2%) was used as the standard anesthetic drug.
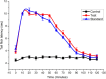
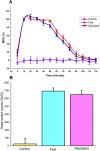
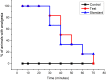
Results: In TFL, Injection Harsha 22 showed anesthetic effects up to 90 minutes after application. Also, the duration of anesthesia in rats that were administered subcutaneously with Injection Harsha 22 was comparable to that of the rats treated with commercial lignocaine (2%). In an electrical stimulation test, single administration of Injection Harsha 22 to rats significantly prolonged analgesia compared with the normal control group. The median duration of analgesia in rats administered subcutaneously with Injection Harsha 22 and lignocaine solution was 40 minutes and 35 minutes, respectively. Furthermore, Injection Harsha 22 does not interfere with the hematopoietic system of the experiment animals.
Conclusion: Thus, the present investigation established the in vivo anesthetic and analgesic potential of Injection Harsha 22 in experimental animals. Hence, it can be concluded that Injection Harsha 22 can become a prominent substitute for lignocaine as a local anaesthetic agent after establishing its efficacy through stringent clinical trials in humans.
Keywords: Wistar rats; analgesic; anesthetic; local anesthetic; polyherbal anesthetic formulation.
© 2023 Sasidharan et al.
Conflict of interest statement
The authors disclosed no conflicts of interest in this work.
Figures

Similar articles
-
Effectiveness of ondansetron as an adjunct to lidocaine intravenous regional anesthesia on tourniquet pain and postoperative pain in patients undergoing elective hand surgery: a systematic review protocol.JBI Database System Rev Implement Rep. 2015 Jan;13(1):27-38. doi: 10.11124/jbisrir-2015-1768. JBI Database System Rev Implement Rep. 2015. PMID: 26447005
-
A model of intravenous regional anesthesia in rats.Anesth Analg. 2010 Apr 1;110(4):1227-32. doi: 10.1213/ANE.0b013e3181ce1f84. Anesth Analg. 2010. PMID: 20357158
-
KEA-1010, a ketamine ester analogue, retains analgesic and sedative potency but is devoid of Psychomimetic effects.BMC Pharmacol Toxicol. 2019 Dec 19;20(1):85. doi: 10.1186/s40360-019-0374-y. BMC Pharmacol Toxicol. 2019. PMID: 31856925 Free PMC article.
-
Anesthetic and analgesic efficacy of bupivacaine in mandibular third molar surgery: a systematic review and meta-analysis.Quintessence Int. 2020;51(7):586-597. doi: 10.3290/j.qi.a44633. Quintessence Int. 2020. PMID: 32500866
-
Bupivacaine versus lignocaine as the choice of locall anesthetic agent for impacted third molar surgery a review.J Pharm Bioallied Sci. 2015 Apr;7(Suppl 1):S230-3. doi: 10.4103/0975-7406.155921. J Pharm Bioallied Sci. 2015. PMID: 26015720 Free PMC article. Review.
Cited by
-
Research Trends of Pharmacopuncture: a bibliometric analysis using VOSviewer (2007-2023).J Pharmacopuncture. 2023 Sep 30;26(3):227-237. doi: 10.3831/KPI.2023.26.3.227. J Pharmacopuncture. 2023. PMID: 37799619 Free PMC article. Review.
-
Impaired Basal Forebrain Cholinergic Neuron GDNF Signaling Contributes to Perioperative Sleep Deprivation-Induced Chronicity of Postsurgical Pain in Mice Through Regulating Cholinergic Neuronal Activity, Apoptosis, and Autophagy.CNS Neurosci Ther. 2024 Dec;30(12):e70147. doi: 10.1111/cns.70147. CNS Neurosci Ther. 2024. PMID: 39639706 Free PMC article.
References
LinkOut - more resources
Full Text Sources

