InterMEL: An international biorepository and clinical database to uncover predictors of survival in early-stage melanoma
- PMID: 37011054
- PMCID: PMC10069769
- DOI: 10.1371/journal.pone.0269324
InterMEL: An international biorepository and clinical database to uncover predictors of survival in early-stage melanoma
Abstract
Introduction: We are conducting a multicenter study to identify classifiers predictive of disease-specific survival in patients with primary melanomas. Here we delineate the unique aspects, challenges, and best practices for optimizing a study of generally small-sized pigmented tumor samples including primary melanomas of at least 1.05mm from AJTCC TNM stage IIA-IIID patients. We also evaluated tissue-derived predictors of extracted nucleic acids' quality and success in downstream testing. This ongoing study will target 1,000 melanomas within the international InterMEL consortium.
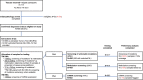
Methods: Following a pre-established protocol, participating centers ship formalin-fixed paraffin embedded (FFPE) tissue sections to Memorial Sloan Kettering Cancer Center for the centralized handling, dermatopathology review and histology-guided coextraction of RNA and DNA. Samples are distributed for evaluation of somatic mutations using next gen sequencing (NGS) with the MSK-IMPACTTM assay, methylation-profiling (Infinium MethylationEPIC arrays), and miRNA expression (Nanostring nCounter Human v3 miRNA Expression Assay).
Results: Sufficient material was obtained for screening of miRNA expression in 683/685 (99%) eligible melanomas, methylation in 467 (68%), and somatic mutations in 560 (82%). In 446/685 (65%) cases, aliquots of RNA/DNA were sufficient for testing with all three platforms. Among samples evaluated by the time of this analysis, the mean NGS coverage was 249x, 59 (18.6%) samples had coverage below 100x, and 41/414 (10%) failed methylation QC due to low intensity probes or insufficient Meta-Mixed Interquartile (BMIQ)- and single sample (ss)- Noob normalizations. Six of 683 RNAs (1%) failed Nanostring QC due to the low proportion of probes above the minimum threshold. Age of the FFPE tissue blocks (p<0.001) and time elapsed from sectioning to co-extraction (p = 0.002) were associated with methylation screening failures. Melanin reduced the ability to amplify fragments of 200bp or greater (absent/lightly pigmented vs heavily pigmented, p<0.003). Conversely, heavily pigmented tumors rendered greater amounts of RNA (p<0.001), and of RNA above 200 nucleotides (p<0.001).
Conclusion: Our experience with many archival tissues demonstrates that with careful management of tissue processing and quality control it is possible to conduct multi-omic studies in a complex multi-institutional setting for investigations involving minute quantities of FFPE tumors, as in studies of early-stage melanoma. The study describes, for the first time, the optimal strategy for obtaining archival and limited tumor tissue, the characteristics of the nucleic acids co-extracted from a unique cell lysate, and success rate in downstream applications. In addition, our findings provide an estimate of the anticipated attrition that will guide other large multicenter research and consortia.
Copyright: This is an open access article, free of all copyright, and may be freely reproduced, distributed, transmitted, modified, built upon, or otherwise used by anyone for any lawful purpose. The work is made available under the Creative Commons CC0 public domain dedication.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: R.A.S. has received fees for professional services from F. Hoffmann-La Roche Ltd, Evaxion, Provectus Biopharmaceuticals Australia, Qbiotics, Novartis, Merck Sharp & Dohme, NeraCare, AMGEN Inc., Bristol-Myers Squibb, Myriad Genetics, GlaxoSmithKline. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures




References
-
- Howlader N, Noone A, Krapcho M, Garshell J, Neyman N, Altekruse S, et al. SEER Cancer Statistics Review Bethesda, MD: National Cancer Institute; 1975–2010 [updated April 2013. Available from: http://seer.cancer.gov/csr/1975_2010/].
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

