Epsin1-mediated exosomal sorting of Dll4 modulates the tubular-macrophage crosstalk in diabetic nephropathy
- PMID: 37016580
- PMCID: PMC10188907
- DOI: 10.1016/j.ymthe.2023.03.027
Epsin1-mediated exosomal sorting of Dll4 modulates the tubular-macrophage crosstalk in diabetic nephropathy
Abstract
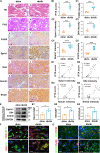
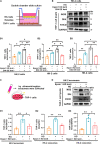
Tubular epithelial cells (TECs) play critical roles in the development of diabetic nephropathy (DN), and can activate macrophages through the secretion of exosomes. However, the mechanism(s) of TEC-exosomes in macrophage activation under DN remains unknown. By mass spectrometry, 1,644 differentially expressed proteins, especially Dll4, were detected in the urine exosomes of DN patients compared with controls, which was confirmed by western blot assay. Elevated Epsin1 and Dll4/N1ICD expression was observed in kidney tissues in both DN patients and db/db mice and was positively associated with tubulointerstitial damage. Exosomes from high glucose (HG)-treated tubular cells (HK-2) with Epsin1 knockdown (KD) ameliorated macrophage activation, TNF-α, and IL-6 expression, and tubulointerstitial damage in C57BL/6 mice in vivo. In an in vitro study, enriched Dll4 was confirmed in HK-2 cells stimulated with HG, which was captured by THP-1 cells and promoted M1 macrophage activation. In addition, Epsin1 modulated the content of Dll4 in TEC-exosomes stimulated with HG. TEC-exosomes with Epsin1-KD significantly inhibited N1ICD activation and iNOS expression in THP-1 cells compared with incubation with HG alone. These findings suggested that Epsin1 could modulate tubular-macrophage crosstalk in DN by mediating exosomal sorting of Dll4 and Notch1 activation.
Keywords: Dll4; Epsin1; diabetic nephropathy; exosome; inflammation.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

