Approaches for the modulation of mechanosensitive MscL channel pores
- PMID: 37021145
- PMCID: PMC10069478
- DOI: 10.3389/fchem.2023.1162412
Approaches for the modulation of mechanosensitive MscL channel pores
Abstract
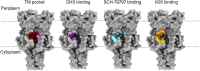
MscL was the first mechanosensitive ion channel identified in bacteria. The channel opens its large pore when the turgor pressure of the cytoplasm increases close to the lytic limit of the cellular membrane. Despite their ubiquity across organisms, their importance in biological processes, and the likelihood that they are one of the oldest mechanisms of sensory activation in cells, the exact molecular mechanism by which these channels sense changes in lateral tension is not fully understood. Modulation of the channel has been key to understanding important aspects of the structure and function of MscL, but a lack of molecular triggers of these channels hindered early developments in the field. Initial attempts to activate mechanosensitive channels and stabilize functionally relevant expanded or open states relied on mutations and associated post-translational modifications that were often cysteine reactive. These sulfhydryl reagents positioned at key residues have allowed the engineering of MscL channels for biotechnological purposes. Other studies have modulated MscL by altering membrane properties, such as lipid composition and physical properties. More recently, a variety of structurally distinct agonists have been shown bind to MscL directly, close to a transmembrane pocket that has been shown to have an important role in channel mechanical gating. These agonists have the potential to be developed further into antimicrobial therapies that target MscL, by considering the structural landscape and properties of these pockets.
Keywords: MscL; agonists; antibiotics; mechanosensitive channel; membrane pores; modulators.
Copyright © 2023 Lane and Pliotas.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Alexander S., Mathie A., Peters J. (2011). Ion channels. Br. J. Pharmacol. 164 (1), 137–174. 10.1111/j.1476-5381.2011.01649_5.x - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

