Novel strategy for oncogenic alteration-induced lipid metabolism reprogramming in pancreatic cancer
- PMID: 37021976
- PMCID: PMC10326418
- DOI: 10.3724/abbs.2023045
Novel strategy for oncogenic alteration-induced lipid metabolism reprogramming in pancreatic cancer
Abstract
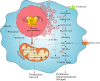
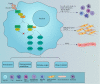
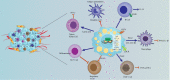
The pathogenesis of pancreatic cancer involves substantial metabolic reprogramming, resulting in abnormal proliferation of tumor cells. This tumorigenic reprogramming is often driven by genetic mutations, such as activating mutations of the KRAS oncogene and inactivating or deletions of the tumor suppressor genes SMAD4, CDKN2A, and TP53, which play a critical role in the initiation and development of pancreatic cancer. As a normal cell gradually develops into a cancer cell, a series of signature characteristics are acquired: activation of signaling pathways that sustain proliferation; an ability to resist growth inhibitory signals and evade apoptosis; and an ability to generate new blood vessels and invade and metastasize. In addition to these features, recent research has revealed that metabolic reprogramming and immune escape are two other novel characteristics of tumor cells. The effect of the interactions between tumor and immune cells on metabolic reprogramming is a key factor determining the antitumor immunotherapy response. Lipid metabolism reprogramming, a feature of many malignancies, not only plays a role in maintaining tumor cell proliferation but also alters the tumor microenvironment by inducing the release of metabolites that in turn affect the metabolism of normal immune cells, ultimately leading to the attenuation of the antitumor immune response and resistance to immunotherapy. Pancreatic cancer has been found to have substantial lipid metabolism reprogramming, but the mechanisms remain elusive. Therefore, this review focuses on the mechanisms regulating lipid metabolism reprogramming in pancreatic cancer cells to provide new therapeutic targets and aid the development of new therapeutic strategies for pancreatic cancer.
Keywords: drug resistance; immune escape; lipid metabolism reprogramming; pancreatic cancer; tumor microenvironment.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
Metabolic reprogramming of immune cells in pancreatic cancer progression.Biomed Pharmacother. 2023 Jan;157:113992. doi: 10.1016/j.biopha.2022.113992. Epub 2022 Nov 14. Biomed Pharmacother. 2023. PMID: 36395610 Review.
-
Reprogramming of lipid metabolism in the tumor microenvironment: a strategy for tumor immunotherapy.Lipids Health Dis. 2024 Feb 1;23(1):35. doi: 10.1186/s12944-024-02024-0. Lipids Health Dis. 2024. PMID: 38302980 Free PMC article. Review.
-
Glucose Metabolism Reprogramming of Immune Cells in the Microenvironment of Pancreatic and Hepatobiliary Cancers.J Gastroenterol Hepatol. 2025 Feb;40(2):355-366. doi: 10.1111/jgh.16873. Epub 2025 Jan 8. J Gastroenterol Hepatol. 2025. PMID: 39780341 Review.
-
Metabolic reprogramming due to hypoxia in pancreatic cancer: Implications for tumor formation, immunity, and more.Biomed Pharmacother. 2021 Sep;141:111798. doi: 10.1016/j.biopha.2021.111798. Epub 2021 Jun 10. Biomed Pharmacother. 2021. PMID: 34120068 Review.
-
The Role of Tumor Metabolic Reprogramming in Tumor Immunity.Int J Mol Sci. 2023 Dec 13;24(24):17422. doi: 10.3390/ijms242417422. Int J Mol Sci. 2023. PMID: 38139250 Free PMC article. Review.
Cited by
-
Exploring the underlying biology of cancer and potential therapeutic strategies: a special issue focused on mechanism-based studies.Acta Biochim Biophys Sin (Shanghai). 2023 Jun 19;55(6):891-892. doi: 10.3724/abbs.2023114. Acta Biochim Biophys Sin (Shanghai). 2023. PMID: 37337636 Free PMC article. No abstract available.
-
miR-373-3p promotes aerobic glycolysis in colon cancer cells by targeting MFN2.Acta Biochim Biophys Sin (Shanghai). 2024 Jun 27;56(10):1498-1508. doi: 10.3724/abbs.2024090. Acta Biochim Biophys Sin (Shanghai). 2024. PMID: 38946424 Free PMC article.
References
-
- Rawla P, Sunkara T, Gaduputi V. Epidemiology of pancreatic cancer: global trends, etiology and risk factors. World J Oncol. . 2019;10:10–27. doi: 10.14740/wjon1166. - DOI - PMC - PubMed
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022, 72: 7–33 - PubMed
-
- Rahib L, Wehner MR, Matrisian LM, Nead KT. Estimated projection of US cancer incidence and death to 2040. JAMA Network Open. 2021, 4: e214708 - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

