All DACs in a Row: Domain Architectures of Bacterial and Archaeal Diadenylate Cyclases
- PMID: 37022175
- PMCID: PMC10127676
- DOI: 10.1128/jb.00023-23
All DACs in a Row: Domain Architectures of Bacterial and Archaeal Diadenylate Cyclases
Abstract
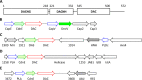
Cyclic dimeric AMP (c-di-AMP) is a widespread second messenger that controls such key functions as osmotic homeostasis, peptidoglycan biosynthesis, and response to various stresses. C-di-AMP is synthesized by diadenylate cyclases that contain the DAC (DisA_N) domain, which was originally characterized as the N-terminal domain in the DNA integrity scanning protein DisA. In other experimentally studied diadenylate cyclases, DAC domain is typically located at the protein C termini and its enzymatic activity is controlled by one or more N-terminal domains. As in other bacterial signal transduction proteins, these N-terminal modules appear to sense environmental or intracellular signals through ligand binding and/or protein-protein interactions. Studies of bacterial and archaeal diadenylate cyclases also revealed numerous sequences with uncharacterized N-terminal regions. This work provides a comprehensive review of the N-terminal domains of bacterial and archaeal diadenylate cyclases, including the description of five previously undefined domains and three PK_C-related domains of the DacZ_N superfamily. These data are used to classify diadenylate cyclases into 22 families, based on their conserved domain architectures and the phylogeny of their DAC domains. Although the nature of the regulatory signals remains obscure, the association of certain dac genes with anti-phage defense CBASS systems and other phage-resistance genes suggests that c-di-AMP might also be involved in the signaling of phage infection.
Keywords: c-di-AMP; diadenylate cyclase; homeostasis; osmotic pressure; phage defense; pyruvate kinase allosteric regulation; sensor domain; signal transduction.
Conflict of interest statement
The author declares no conflict of interest.
Figures


Similar articles
-
Replenishing the cyclic-di-AMP pool: regulation of diadenylate cyclase activity in bacteria.Curr Genet. 2016 Nov;62(4):731-738. doi: 10.1007/s00294-016-0600-8. Epub 2016 Apr 13. Curr Genet. 2016. PMID: 27074767 Review.
-
Control of the diadenylate cyclase CdaS in Bacillus subtilis: an autoinhibitory domain limits cyclic di-AMP production.J Biol Chem. 2014 Jul 25;289(30):21098-107. doi: 10.1074/jbc.M114.562066. Epub 2014 Jun 16. J Biol Chem. 2014. PMID: 24939848 Free PMC article.
-
Great times for small molecules: c-di-AMP, a second messenger candidate in Bacteria and Archaea.Sci Signal. 2008 Aug 19;1(33):pe39. doi: 10.1126/scisignal.133pe39. Sci Signal. 2008. PMID: 18714086
-
Making and Breaking of an Essential Poison: the Cyclases and Phosphodiesterases That Produce and Degrade the Essential Second Messenger Cyclic di-AMP in Bacteria.J Bacteriol. 2018 Dec 7;201(1):e00462-18. doi: 10.1128/JB.00462-18. Print 2019 Jan 1. J Bacteriol. 2018. PMID: 30224435 Free PMC article. Review.
-
Structural analysis of the diadenylate cyclase reaction of DNA-integrity scanning protein A (DisA) and its inhibition by 3'-dATP.Biochem J. 2015 Aug 1;469(3):367-74. doi: 10.1042/BJ20150373. Epub 2015 May 27. Biochem J. 2015. PMID: 26014055
Cited by
-
The many roles of cyclic di-AMP to control the physiology of Bacillus subtilis.Microlife. 2023 Oct 20;4:uqad043. doi: 10.1093/femsml/uqad043. eCollection 2023. Microlife. 2023. PMID: 37954098 Free PMC article. Review.
-
Putative nucleotide-based second messengers in archaea.Microlife. 2023 Jun 5;4:uqad027. doi: 10.1093/femsml/uqad027. eCollection 2023. Microlife. 2023. PMID: 37305433 Free PMC article. Review.
-
Cyclic-di-AMP modulates cellular turgor in response to defects in bacterial cell wall synthesis.Nat Microbiol. 2025 Jul;10(7):1698-1710. doi: 10.1038/s41564-025-02027-2. Epub 2025 Jun 17. Nat Microbiol. 2025. PMID: 40528006 Free PMC article.
-
COG database update 2024.Nucleic Acids Res. 2025 Jan 6;53(D1):D356-D363. doi: 10.1093/nar/gkae983. Nucleic Acids Res. 2025. PMID: 39494517 Free PMC article.
-
ComFB, a new widespread family of c-di-NMP receptor proteins.bioRxiv [Preprint]. 2024 Nov 10:2024.11.10.622515. doi: 10.1101/2024.11.10.622515. bioRxiv. 2024. PMID: 39574629 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

