Glioblastomas: Hijacking Metabolism to Build a Flexible Shield for Therapy Resistance
- PMID: 37022791
- PMCID: PMC10655009
- DOI: 10.1089/ars.2022.0088
Glioblastomas: Hijacking Metabolism to Build a Flexible Shield for Therapy Resistance
Abstract
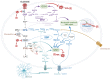
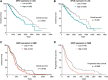
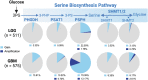
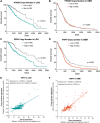
Significance: Glioblastomas (GBMs) are among the most lethal tumors despite the almost exclusive localization to the brain. This is largely due to therapeutic resistance. Radiation and chemotherapy significantly increase the survival for GBM patients, however, GBMs always recur, and the median overall survival is just over a year. Proposed reasons for such intractable resistance to therapy are numerous and include tumor metabolism, in particular, the ability of tumor cells to reconfigure metabolic fluxes on demand (metabolic plasticity). Understanding how the hard-wired, oncogene-driven metabolic tendencies of GBMs intersect with flexible, context-induced metabolic rewiring promises to reveal novel approaches for combating therapy resistance. Recent Advances: Personalized genome-scale metabolic flux models have recently provided evidence that metabolic flexibility promotes radiation resistance in cancer and identified tumor redox metabolism as a major predictor for resistance to radiation therapy (RT). It was demonstrated that radioresistant tumors, including GBM, reroute metabolic fluxes to boost the levels of reducing factors of the cell, thus enhancing clearance of reactive oxygen species that are generated during RT and promoting survival. Critical Issues: The current body of knowledge from published studies strongly supports the notion that robust metabolic plasticity can act as a (flexible) shield against the cytotoxic effects of standard GBM therapies, thus driving therapy resistance. The limited understanding of the critical drivers of such metabolic plasticity hampers the rational design of effective combination therapies. Future Directions: Identifying and targeting regulators of metabolic plasticity, rather than specific metabolic pathways, in combination with standard-of-care treatments have the potential to improve therapeutic outcomes in GBM. Antioxid. Redox Signal. 39, 957-979.
Keywords: glioblastoma; metabolism; plasticity; redox.
Conflict of interest statement
The authors have no conflict of interest.
Figures


Similar articles
-
Combined acetyl-11-keto-β-boswellic acid and radiation treatment inhibited glioblastoma tumor cells.PLoS One. 2018 Jul 3;13(7):e0198627. doi: 10.1371/journal.pone.0198627. eCollection 2018. PLoS One. 2018. PMID: 29969452 Free PMC article.
-
Increased cure rate of glioblastoma using concurrent therapy with radiotherapy and arsenic trioxide.Int J Radiat Oncol Biol Phys. 2004 Sep 1;60(1):197-203. doi: 10.1016/j.ijrobp.2004.02.013. Int J Radiat Oncol Biol Phys. 2004. PMID: 15337556
-
Molecular Markers of Therapy-Resistant Glioblastoma and Potential Strategy to Combat Resistance.Int J Mol Sci. 2018 Jun 14;19(6):1765. doi: 10.3390/ijms19061765. Int J Mol Sci. 2018. PMID: 29899215 Free PMC article. Review.
-
The Importance of Tumor Stem Cells in Glioblastoma Resistance to Therapy.Int J Mol Sci. 2021 Apr 8;22(8):3863. doi: 10.3390/ijms22083863. Int J Mol Sci. 2021. PMID: 33917954 Free PMC article. Review.
-
Antiangiogenic therapies in glioblastoma multiforme.Expert Rev Anticancer Ther. 2012 May;12(5):643-54. doi: 10.1586/era.12.35. Expert Rev Anticancer Ther. 2012. PMID: 22594899 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

