A novel immune checkpoint score system for prognostic evaluation in pancreatic adenocarcinoma
- PMID: 37024802
- PMCID: PMC10080823
- DOI: 10.1186/s12876-023-02748-w
A novel immune checkpoint score system for prognostic evaluation in pancreatic adenocarcinoma
Abstract
Background: Pancreatic adenocarcinoma (PAAD) remains a lethal malignancy making the detection of novel prognostic biomarkers urgent. Limited studies have investigated the predictive capability of immune checkpoints in PAAD.
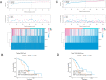
Method: Gene expression data and correlative clinical information of PAAD cohort were obtained from public databases, including TCGA, ICGC, GTEX and GEO databases. Risk factors were screened and used to establish a risk score model through LASSO and Cox regression analyses. The prognostic ability of the risk score model was demonstrated. The association between risk score with immune cells infiltration, immune checkpoint genes expression, immunogenic cell death, somatic mutations and signaling pathways enrichment were analysed. scRNA-seq data were collected to confirmed the immune checkpoints expression in PAAD samples. The prognosis prediction ability of OX40/TNFRSF4 was identified. The mRNA and protein expression of OX40 in our clinical specimens were examined by RT-PCR and IHC method and its prognosis ability was verified.
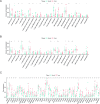
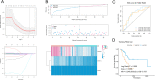
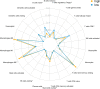
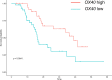
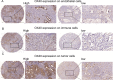
Results: First of all, the difference of immune microenvironment between pancreatic cancer and adjacent tissues was shown. A risk score system based on three immune checkpoints (OX40, TNFSF14 and KIR3DL1) was established. The risk score model was an independent prognostic factor and performed well regarding overall survival (OS) predictions among PAAD patients. A nomogram was established to facilitate the risk model application in clinical prognosis. Immune cells including naive B cells, CD8+ T cells and Tregs were negatively correlated with the risk score. The risk score was associated with expression of immune checkpoint genes, immunogenic cell death related genes and somatic mutations. Glycolysis processes, IL-2-STAT5, IL-6-STAT3, and mTORC1 signaling pathways were enriched in the high-risk score group. Furthermore, scRNA-seq data confirmed that TNFRSF4, TNFSF14 and KIR3DL1 were expressed on immune cells in PAAD samples. We then identified OX40 as an independent prognosis-related gene, and a higher OX40 expression was associated with increased survival rate and immune environment change. In 84 PAAD clinical specimens collected from our center, we confirmed that higher OX40 mRNA expression levels were related to a good prognosis. The protein expression of OX40 on tumor-infiltrating immune cells (TIICs), endothelial cells and tumor cells was verified in PAAD tissues by immunohistochemistry (IHC) method.
Conclusions: Overall, our findings strongly suggested that the three-immune checkpoints score system might be useful in the prognosis and design of personalized treatments for PAAD patients. Finally, we identified OX40 as an independent potential biomarker for PAAD prognosis prediction.
Keywords: Immune checkpoints; Immune risk score model; OX40; Pancreatic adenocarcinoma; Prognostic evaluation.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
MeSH terms
Grants and funding
- 82072693, 81902417, 82172884/the National Natural Science Foundation of China
- 2019-01-07-00-07-E00057/the Scientific Innovation Project of Shanghai Education Committee
- SHDC12018109/Clinical and Scientific Innovation Project of Shanghai Hospital Development Center
- SHDC2020CR1006A/Clinical Research Plan of Shanghai Hospital Development Center
- 2020YFA0803202/National Key Research and Development Program of China
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

