Cooperative NF-κB and Notch1 signaling promotes macrophage-mediated MenaINV expression in breast cancer
- PMID: 37024946
- PMCID: PMC10080980
- DOI: 10.1186/s13058-023-01628-1
Cooperative NF-κB and Notch1 signaling promotes macrophage-mediated MenaINV expression in breast cancer
Abstract
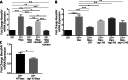
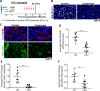
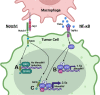
Metastasis is a multistep process that leads to the formation of clinically detectable tumor foci at distant organs and frequently to patient demise. Only a subpopulation of breast cancer cells within the primary tumor can disseminate systemically and cause metastasis. To disseminate, cancer cells must express MenaINV, an isoform of the actin regulatory protein Mena, encoded by the ENAH gene, that endows tumor cells with transendothelial migration activity, allowing them to enter and exit the blood circulation. We have previously demonstrated that MenaINV mRNA and protein expression is induced in cancer cells by macrophage contact. In this study, we discovered the precise mechanism by which macrophages induce MenaINV expression in tumor cells. We examined the promoter of the human and mouse ENAH gene and discovered a conserved NF-κB transcription factor binding site. Using live imaging of an NF-κB activity reporter and staining of fixed tissues from mouse and human breast cancer, we further determined that for maximal induction of MenaINV in cancer cells, NF-κB needs to cooperate with the Notch1 signaling pathway. Mechanistically, Notch1 signaling does not directly increase MenaINV expression, but it enhances and sustains NF-κB signaling through retention of p65, an NF-κB transcription factor, in the nucleus of tumor cells, leading to increased MenaINV expression. In mice, these signals are augmented following chemotherapy treatment and abrogated upon macrophage depletion. Targeting Notch1 signaling in vivo decreased NF-κB signaling activation and MenaINV expression in the primary tumor and decreased metastasis. Altogether, these data uncover mechanistic targets for blocking MenaINV induction that should be explored clinically to decrease cancer cell dissemination and improve survival of patients with metastatic disease.
Keywords: Breast cancer; MenaINV; NF-κB; Notch1; TMEM doorways.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Update of
-
Cooperative NF-κB and Notch1 signaling promotes macrophage-mediated MenaINV expression in breast cancer.bioRxiv [Preprint]. 2023 Jan 3:2023.01.03.522642. doi: 10.1101/2023.01.03.522642. bioRxiv. 2023. Update in: Breast Cancer Res. 2023 Apr 6;25(1):37. doi: 10.1186/s13058-023-01628-1. PMID: 36711751 Free PMC article. Updated. Preprint.
Similar articles
-
Cooperative NF-κB and Notch1 signaling promotes macrophage-mediated MenaINV expression in breast cancer.bioRxiv [Preprint]. 2023 Jan 3:2023.01.03.522642. doi: 10.1101/2023.01.03.522642. bioRxiv. 2023. Update in: Breast Cancer Res. 2023 Apr 6;25(1):37. doi: 10.1186/s13058-023-01628-1. PMID: 36711751 Free PMC article. Updated. Preprint.
-
Paclitaxel causes de novo induction of invasive breast cancer cells by repolarizing tumor-associated macrophages.bioRxiv [Preprint]. 2025 Jun 8:2025.01.13.632767. doi: 10.1101/2025.01.13.632767. bioRxiv. 2025. PMID: 39868194 Free PMC article. Preprint.
-
Invasive breast carcinoma cells from patients exhibit MenaINV- and macrophage-dependent transendothelial migration.Sci Signal. 2014 Nov 25;7(353):ra112. doi: 10.1126/scisignal.2005329. Sci Signal. 2014. PMID: 25429076 Free PMC article.
-
Macrophage-dependent tumor cell transendothelial migration is mediated by Notch1/MenaINV-initiated invadopodium formation.Sci Rep. 2016 Nov 30;6:37874. doi: 10.1038/srep37874. Sci Rep. 2016. PMID: 27901093 Free PMC article.
-
Coexpression of Notch1 and NF-kappaB signaling pathway components in human cervical cancer progression.Gynecol Oncol. 2007 Feb;104(2):352-61. doi: 10.1016/j.ygyno.2006.08.054. Epub 2006 Nov 13. Gynecol Oncol. 2007. PMID: 17098279
Cited by
-
NOTCH1 combined with chemotherapy synergistically inhibits triple-negative breast cancer.World J Clin Oncol. 2025 Jun 24;16(6):106197. doi: 10.5306/wjco.v16.i6.106197. World J Clin Oncol. 2025. PMID: 40585842 Free PMC article.
-
Diverse roles of UBE2S in cancer and therapy resistance: Biological functions and mechanisms.Heliyon. 2024 Jan 18;10(2):e24465. doi: 10.1016/j.heliyon.2024.e24465. eCollection 2024 Jan 30. Heliyon. 2024. PMID: 38312603 Free PMC article. Review.
-
Targeting CSF-1 signaling between tumor cells and macrophages at TMEM doorways inhibits breast cancer dissemination.Oncogene. 2025 Jul 11. doi: 10.1038/s41388-025-03485-y. Online ahead of print. Oncogene. 2025. PMID: 40646332
-
New insights into the anticancer effects of Polycladia crinita aqueous extract and its selenium nanoformulation against the solid Ehrlich carcinoma model in mice via VEGF, notch 1, NF-кB, cyclin D1, and caspase 3 signaling pathway.Front Pharmacol. 2024 Feb 16;15:1345516. doi: 10.3389/fphar.2024.1345516. eCollection 2024. Front Pharmacol. 2024. PMID: 38469406 Free PMC article.
-
Design, synthesis and evaluation of pyrrolobenzodiazepine (PBD)-based PROTAC conjugates for the selective degradation of the NF-κB RelA/p65 subunit.RSC Med Chem. 2025 May 8. doi: 10.1039/d5md00316d. Online ahead of print. RSC Med Chem. 2025. PMID: 40443648 Free PMC article.
References
-
- Karagiannis GS, Poutahidis T, Erdman SE, Kirsch R, Riddell RH, Diamandis EP. Cancer-associated fibroblasts drive the progression of metastasis through both paracrine and mechanical pressure on cancer tissue. Mol Cancer Res. 2012;10(11):1403–1418. doi: 10.1158/1541-7786.MCR-12-0307. - DOI - PMC - PubMed
-
- Zhu Y, Knolhoff BL, Meyer MA, Nywening TM, West BL, Luo J, et al. CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res. 2014;74(18):5057–5069. doi: 10.1158/0008-5472.CAN-13-3723. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

