Hyperthermic intravesical chemotherapy with mitomycin-C for the treatment of high-risk non-muscle-invasive bladder cancer patients
- PMID: 37025474
- PMCID: PMC10071075
- DOI: 10.1002/bco2.203
Hyperthermic intravesical chemotherapy with mitomycin-C for the treatment of high-risk non-muscle-invasive bladder cancer patients
Abstract
Objectives: The objectives of the study are to explore tolerability, acceptability and oncological outcomes for patients with high-risk non-muscle-invasive bladder cancer (NMIBC) treated with hyperthermic intravesical chemotherapy (HIVEC) and mitomycin-C (MMC) at our institution.
Patients and methods: Our single-institution, observational study consists of consecutive high-risk NMIBC patients treated with HIVEC and MMC. Our HIVEC protocol included six weekly instillations (induction), followed by two further cycles of three instillations (maintenance) (6 + 3 + 3) if there was cystoscopic response. Patient demographics, instillation dates and adverse events (AEs) were collected prospectively in our dedicated HIVEC clinic. Retrospective case-note review was performed to evaluate oncological outcomes. Primary outcomes were tolerability and acceptability of HIVEC protocol; secondary outcomes were 12-month recurrence-free, progression-free and overall survival.
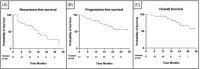
Results: In total, 57 patients (median age 80.3 years) received HIVEC and MMC, with a median follow-up of 18 months. Of these, 40 (70.2%) had recurrent tumours, and 29 (50.9%) had received prior Bacillus Calmette-Guérin (BCG). HIVEC induction was completed by 47 (82.5%) patients, but only 19 (33.3%) completed the full protocol. Disease recurrence (28.9%) and AEs (28.9%) were the most common reasons for incompletion of protocol; five (13.2%) patients stopped treatment due to logistical challenges. AEs occurred in 20 (35.1%) patients; the most frequently documented were rash (10.5%), urinary tract infection (8.8%) and bladder spasm (8.8%). Progression during treatment occurred in 11 (19.3%) patients, 4 (7.0%) of whom had muscle invasion and 5 (8.8%) subsequently required radical treatment. Patients who had received prior BCG were significantly more likely to progress (p = 0.04). 12-month recurrence-free, progression-free and overall survival rates were 67.5%, 82.2%, and 94.7%, respectively.
Conclusions: Our single-institution experience suggests that HIVEC and MMC are tolerable and acceptable. Oncological outcomes in this predominantly elderly, pretreated cohort are promising; however, disease progression was higher in patients pretreated with BCG. Further randomised noninferiority trials comparing HIVEC versus BCG in high-risk NMIBC are required.
Keywords: acceptability; high risk; hyperthermic intravesical chemotherapy; intravesical treatment; mitomycin C; non‐muscle‐invasive bladder cancer; tolerability.
© 2022 The Authors. BJUI Compass published by John Wiley & Sons Ltd on behalf of BJU International Company.
Conflict of interest statement
There are no conflicts of interest to disclose.
Figures
References
-
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin [Internet]. 2021. May 1 [cited 2022 Apr 6];71(3):209–49. Available from: https://onlinelibrary.wiley.com/doi/full/10.3322/caac.21660 - DOI - PubMed
-
- Cumberbatch MGK, Jubber I, Black PC, Esperto F, Figueroa JD, Kamat AM, et al. Epidemiology of bladder cancer: a systematic review and contemporary update of risk factors in 2018. Eur Urol [Internet]. 2018. Dec 1 [cited 2019 Feb 3];74(6):784–95. Available from: https://www.sciencedirect.com/science/article/pii/S0302283818306511#bib0525 - PubMed
-
- Sylvester RJ, van der Meijden APM, Oosterlinck W, Witjes JA, Bouffioux C, Denis L, et al. Predicting recurrence and progression in individual patients with stage ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol [Internet]. 2006. Mar 1 [cited 2019 Feb 4];49(3):466–77. Available from: https://www.sciencedirect.com/science/article/pii/S0302283805008523?via%... - PubMed
-
- Leal J, Luengo‐Fernandez R, Sullivan R, Witjes JA. Economic burden of bladder cancer across the European Union. Eur Urol [Internet]. 2016. Mar [cited 2020 Jan 20];69(3):438–47. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26508308 - PubMed
LinkOut - more resources
Full Text Sources