Minimal Out-of-Equilibrium Metabolism for Synthetic Cells: A Membrane Perspective
- PMID: 37027340
- PMCID: PMC10127287
- DOI: 10.1021/acssynbio.3c00062
Minimal Out-of-Equilibrium Metabolism for Synthetic Cells: A Membrane Perspective
Abstract
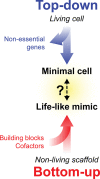
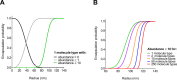
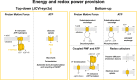
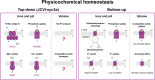
Life-like systems need to maintain a basal metabolism, which includes importing a variety of building blocks required for macromolecule synthesis, exporting dead-end products, and recycling cofactors and metabolic intermediates, while maintaining steady internal physical and chemical conditions (physicochemical homeostasis). A compartment, such as a unilamellar vesicle, functionalized with membrane-embedded transport proteins and metabolic enzymes encapsulated in the lumen meets these requirements. Here, we identify four modules designed for a minimal metabolism in a synthetic cell with a lipid bilayer boundary: energy provision and conversion, physicochemical homeostasis, metabolite transport, and membrane expansion. We review design strategies that can be used to fulfill these functions with a focus on the lipid and membrane protein composition of a cell. We compare our bottom-up design with the equivalent essential modules of JCVI-syn3a, a top-down genome-minimized living cell with a size comparable to that of large unilamellar vesicles. Finally, we discuss the bottlenecks related to the insertion of a complex mixture of membrane proteins into lipid bilayers and provide a semiquantitative estimate of the relative surface area and lipid-to-protein mass ratios (i.e., the minimal number of membrane proteins) that are required for the construction of a synthetic cell.
Keywords: JCVI-syn3a; bottom-up synthetic cells; energy conservation; membrane composition; metabolite transport; minimal metabolism; out-of-equilibrium; physicochemical homeostasis.
Conflict of interest statement
The authors declare no competing financial interest.
Figures





Similar articles
-
Synthesizing artificial cells from giant unilamellar vesicles: state-of-the art in the development of microfluidic technology.Bioessays. 2012 Nov;34(11):992-1001. doi: 10.1002/bies.201200105. Epub 2012 Aug 24. Bioessays. 2012. PMID: 22926929 Review.
-
Exploring Giant Unilamellar Vesicle Production for Artificial Cells - Current Challenges and Future Directions.Small Methods. 2023 Dec;7(12):e2300416. doi: 10.1002/smtd.202300416. Epub 2023 Jul 18. Small Methods. 2023. PMID: 37464561 Review.
-
Biopores/membrane proteins in synthetic polymer membranes.Biochim Biophys Acta Biomembr. 2017 Apr;1859(4):619-638. doi: 10.1016/j.bbamem.2016.10.015. Epub 2016 Oct 29. Biochim Biophys Acta Biomembr. 2017. PMID: 27984019 Review.
-
Artificial Receptor in Synthetic Cells Performs Transmembrane Activation of Proteolysis.Adv Biol (Weinh). 2025 May;9(5):e2400053. doi: 10.1002/adbi.202400053. Epub 2024 May 20. Adv Biol (Weinh). 2025. PMID: 38767247 Free PMC article.
-
Reconstitution of proteins on electroformed giant unilamellar vesicles.Methods Cell Biol. 2015;128:319-38. doi: 10.1016/bs.mcb.2015.02.004. Epub 2015 Apr 8. Methods Cell Biol. 2015. PMID: 25997355 Free PMC article.
Cited by
-
Nucleated synthetic cells with genetically driven intercompartment communication.Proc Natl Acad Sci U S A. 2024 Sep 3;121(36):e2404790121. doi: 10.1073/pnas.2404790121. Epub 2024 Aug 26. Proc Natl Acad Sci U S A. 2024. PMID: 39186653 Free PMC article.
-
Exploring Emergent Properties in Enzymatic Reaction Networks: Design and Control of Dynamic Functional Systems.Chem Rev. 2024 Mar 13;124(5):2553-2582. doi: 10.1021/acs.chemrev.3c00681. Epub 2024 Mar 4. Chem Rev. 2024. PMID: 38476077 Free PMC article. Review.
-
Membrane-embedded CdaA is required for efficient synthesis of second messenger cyclic di-AMP.Commun Biol. 2024 Dec 30;7(1):1710. doi: 10.1038/s42003-024-07420-x. Commun Biol. 2024. PMID: 39739009 Free PMC article.
-
Synthetic syntrophy for adenine nucleotide cross-feeding between metabolically active nanoreactors.Nat Nanotechnol. 2025 Jan;20(1):112-120. doi: 10.1038/s41565-024-01811-1. Epub 2024 Oct 21. Nat Nanotechnol. 2025. PMID: 39433918 Free PMC article.
-
Balancing Permeability and Stability: A Study of Hybrid Membranes for Synthetic Cells Using Lipids and PBd-b-PEO Block Copolymers.Biomacromolecules. 2025 May 12;26(5):2868-2881. doi: 10.1021/acs.biomac.4c01651. Epub 2025 Apr 8. Biomacromolecules. 2025. PMID: 40197008 Free PMC article.
References
-
- Breuer M.; Earnest T. M; Merryman C.; Wise K. S; Sun L.; Lynott M. R; Hutchison C. A; Smith H. O; Lapek J. D; Gonzalez D. J; de Crecy-Lagard V.; Haas D.; Hanson A. D; Labhsetwar P.; Glass J. I; Luthey-Schulten Z. Essential Metabolism for a Minimal Cell. Elife 2019, 8, e36842.10.7554/eLife.36842. - DOI - PMC - PubMed
-
- Reuß D. R.; Altenbuchner J.; Mäder U.; Rath H.; Ischebeck T.; Sappa P. K.; Thürmer A.; Guérin C.; Nicolas P.; Steil L.; Zhu B.; Feussner I.; Klumpp S.; Daniel R.; Commichau F. M.; Völker U.; Stülke J. Large-Scale Reduction of the Bacillus Subtilis Genome: Consequences for the Transcriptional Network, Resource Allocation, and Metabolism. Genome Res. 2017, 27 (2), 289–299. 10.1101/gr.215293.116. - DOI - PMC - PubMed
-
- Hashimoto M.; Ichimura T.; Mizoguchi H.; Tanaka K.; Fujimitsu K.; Keyamura K.; Ote T.; Yamakawa T.; Yamazaki Y.; Mori H.; Katayama T.; Kato J. I. Cell Size and Nucleoid Organization of Engineered Escherichia Coli Cells with a Reduced Genome. Mol. Microbiol. 2005, 55 (1), 137–149. 10.1111/j.1365-2958.2004.04386.x. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

