Methacrylated human recombinant collagen peptide as a hydrogel for manipulating and monitoring stiffness-related cardiac cell behavior
- PMID: 37035009
- PMCID: PMC10074202
- DOI: 10.1016/j.isci.2023.106423
Methacrylated human recombinant collagen peptide as a hydrogel for manipulating and monitoring stiffness-related cardiac cell behavior
Abstract
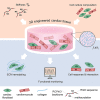
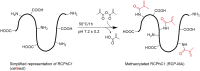
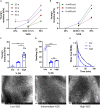
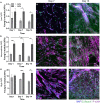
Environmental stiffness is a crucial determinant of cell function. There is a long-standing quest for reproducible and (human matrix) bio-mimicking biomaterials with controllable mechanical properties to unravel the relationship between stiffness and cell behavior. Here, we evaluate methacrylated human recombinant collagen peptide (RCPhC1-MA) hydrogels as a matrix to control 3D microenvironmental stiffness and monitor cardiac cell response. We show that RCPhC1-MA can form hydrogels with reproducible stiffness in the range of human developmental and adult myocardium. Cardiomyocytes (hPSC-CMs) and cardiac fibroblasts (cFBs) remain viable for up to 14 days inside RCPhC1-MA hydrogels while the effect of hydrogel stiffness on extracellular matrix production and hPSC-CM contractility can be monitored in real-time. Interestingly, whereas the beating behavior of the hPSC-CM monocultures is affected by environmental stiffness, this effect ceases when cFBs are present. Together, we demonstrate RCPhC1-MA to be a promising candidate to mimic and control the 3D biomechanical environment of cardiac cells.
Keywords: Biomaterials; Cell biology; Materials in biotechnology; Stem cells research.
© 2023 The Author(s).
Conflict of interest statement
H.A. van Boxtel is an employee of Fujifilm Manufacturing Europe B.V. The results of this study were not influenced by him or any other employee of Fujifilm Manufacturing Europe B.V.
Figures


Similar articles
-
Reversible dynamic mechanics of hydrogels for regulation of cellular behavior.Acta Biomater. 2021 Dec;136:88-98. doi: 10.1016/j.actbio.2021.09.032. Epub 2021 Sep 23. Acta Biomater. 2021. PMID: 34563721 Free PMC article.
-
Bioengineered 3D brain tumor model to elucidate the effects of matrix stiffness on glioblastoma cell behavior using PEG-based hydrogels.Mol Pharm. 2014 Jul 7;11(7):2115-25. doi: 10.1021/mp5000828. Epub 2014 Apr 29. Mol Pharm. 2014. PMID: 24712441
-
Contractile force generation by 3D hiPSC-derived cardiac tissues is enhanced by rapid establishment of cellular interconnection in matrix with muscle-mimicking stiffness.Biomaterials. 2017 Jul;131:111-120. doi: 10.1016/j.biomaterials.2017.03.039. Epub 2017 Mar 30. Biomaterials. 2017. PMID: 28384492 Free PMC article.
-
Mechanosensing of cells in 3D gel matrices based on natural and synthetic materials.Cell Biol Int. 2014 Nov;38(11):1233-43. doi: 10.1002/cbin.10325. Epub 2014 Aug 27. Cell Biol Int. 2014. PMID: 24919784 Review.
-
Native cardiac environment and its impact on engineering cardiac tissue.Biomater Sci. 2019 Aug 20;7(9):3566-3580. doi: 10.1039/c8bm01348a. Biomater Sci. 2019. PMID: 31338495 Review.
Cited by
-
Collagen and Its Derivatives Serving Biomedical Purposes: A Review.Polymers (Basel). 2024 Sep 22;16(18):2668. doi: 10.3390/polym16182668. Polymers (Basel). 2024. PMID: 39339133 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

