Venetoclax plus hypomethylating agents versus intensive chemotherapy for hematological relapse of myeloid malignancies after allo-HSCT
- PMID: 37035180
- PMCID: PMC10079210
- DOI: 10.3389/fonc.2023.1137175
Venetoclax plus hypomethylating agents versus intensive chemotherapy for hematological relapse of myeloid malignancies after allo-HSCT
Abstract
Introduction: Since allogeneic stem cell transplantation (allo-HSCT) is considered one of the curative treatments for acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), hematological relapse following allo-HSCT remained a crucial concern for patients' survival.
Methods: We retrospectively compared patients who received venetoclax plus hypomethylating agents (VEN+HMA, n=23) or intensive chemotherapy (IC, n=42) for hematological relapse of myeloid malignancies after allo-HSCT. HMA selection included decitabine (n=2) and azacitidine (n=21), and combined donor lymphocyte infusion was administered to 21 and 42 patients in VEN+HMA and IC groups, respectively.
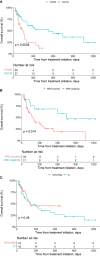
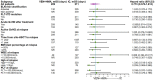
Results: Median age of all patients was 39 (16-64) years old. Overall response rates, including complete response (CR), CR with incomplete recovery of normal neutrophil or platelet counts (CRi) and partial response (PR), were not significantly different between VEN+HMA and IC groups (60.1% versus 64.3%, P=0.785). CR/CRi rate was 52.2% in VEN+HMA and 59.5% in IC group (P=0.567). The rate of relapse after response was 66.7% in VEN+HMA group and 40.7% in IC group (P=0.176). Median overall survival was 209.0 (95%CI 130.9-287.1) days for VEN+HMA group versus 211.0 (95%CI 28.7-393.3) days for IC group (P=0.491). The incidence of lung infection (17.4% versus 50.0%, P=0.010), thrombocytopenia (73.9% versus 95.2%, P=0.035) and acute graft-versus-host disease (aGvHD) (50.0% versus 13.0%, P=0.003) was significantly higher in IC group.
Discussion: In conclusion, VEN+HMA is not inferior to IC regimen in terms of improving response and survival, and is associated with a lower incidence of adverse events and aGvHD. However, further research is required to enhance long-term survival.
Keywords: acute myeloid leukemia; allo-HSCT; myeloid malignancy; relapse; venetoclax.
Copyright © 2023 Chen, Zhen, Zhang, Shen, Pang, Yang, Zhang, Ma, He, Wei, Zhai, Chen, Jiang, Han and Feng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Efficacy and safety of hypomethylating agents in the treatment of AML/MDS patients relapsed post allogenetic hematopoietic stem cell transplantation.Front Oncol. 2024 Dec 9;14:1465334. doi: 10.3389/fonc.2024.1465334. eCollection 2024. Front Oncol. 2024. PMID: 39717745 Free PMC article.
-
Low-dose decitabine plus venetoclax is safe and effective as post-transplant maintenance therapy for high-risk acute myeloid leukemia and myelodysplastic syndrome.Cancer Sci. 2021 Sep;112(9):3636-3644. doi: 10.1111/cas.15048. Epub 2021 Jul 21. Cancer Sci. 2021. PMID: 34185931 Free PMC article.
-
Outcome after allogeneic hematopoietic stem cell transplantation following Venetoclax-based therapy among AML and MDS patients.Ann Hematol. 2022 Dec;101(12):2731-2741. doi: 10.1007/s00277-022-04983-9. Epub 2022 Nov 1. Ann Hematol. 2022. PMID: 36318288
-
Treatment outcomes for newly diagnosed, treatment-naïve TP53-mutated acute myeloid leukemia: a systematic review and meta-analysis.J Hematol Oncol. 2023 Mar 6;16(1):19. doi: 10.1186/s13045-023-01417-5. J Hematol Oncol. 2023. PMID: 36879351 Free PMC article.
-
Impact of treatments before allogeneic hematopoietic stem cell transplantation in patients with higher-risk myelodysplastic syndrome.Leuk Res. 2023 Jan;124:106997. doi: 10.1016/j.leukres.2022.106997. Epub 2022 Dec 7. Leuk Res. 2023. PMID: 36502583 Review.
Cited by
-
Novel Approaches to Treatment of Acute Myeloid Leukemia Relapse Post Allogeneic Stem Cell Transplantation.Int J Mol Sci. 2023 Oct 9;24(19):15019. doi: 10.3390/ijms241915019. Int J Mol Sci. 2023. PMID: 37834466 Free PMC article. Review.
-
Glutathione promotes the synergistic effects of venetoclax and azacytidine against myelodysplastic syndrome‑refractory anemia by regulating the cell cycle.Exp Ther Med. 2023 Oct 26;26(6):574. doi: 10.3892/etm.2023.12274. eCollection 2023 Dec. Exp Ther Med. 2023. PMID: 38023359 Free PMC article.
References
-
- Koreth J, Schlenk R, Kopecky KJ, Honda S, Sierra J, Djulbegovic BJ, et al. . Allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission: systematic review and meta-analysis of prospective clinical trials. JAMA (2009) 301(22):2349–61. doi: 10.1001/jama.2009.813 - DOI - PMC - PubMed
-
- Schmid C, Labopin M, Nagler A, Bornhauser M, Finke J, Fassas A, et al. . Donor lymphocyte infusion in the treatment of first hematological relapse after allogeneic stem-cell transplantation in adults with acute myeloid leukemia: a retrospective risk factors analysis and comparison with other strategies by the EBMT acute leukemia working party. J Clin Oncol (2007) 25(31):4938–45. doi: 10.1200/JCO.2007.11.6053 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

