Modified xiaoyao san combined with chemotherapy for breast cancer: A systematic review and meta-analysis of randomized controlled trials
- PMID: 37035186
- PMCID: PMC10073574
- DOI: 10.3389/fonc.2023.1050337
Modified xiaoyao san combined with chemotherapy for breast cancer: A systematic review and meta-analysis of randomized controlled trials
Abstract
Background: Breast cancer is a common cause of cancer-related death worldwide. Chemotherapy plays an indispensable role in the conventional treatment of breast cancer, bringing some physical burdens and discomfort on cancer patients. Consequently, more and more patients turn to seeking the help of Complementary and Alternative Medicine (CAM), mainly traditional Chinese medicine (TCM). Xiaoyao san (XYS), a classical formula, has been shown to improve symptoms of breast cancer. An increasing number of researches suggest that compared to chemotherapy alone, Chinese herbal medicine combined with chemotherapy could increase effectiveness and reduce toxicity caused by chemotherapy. Emerging experimental research continuously demonstrated some of the components in XYS could stop breast cancer tumor cells from growing. However, the efficacy and safety of modified XYS combined with chemotherapy remain to be determined. Therefore, it is essential to evaluate the comparative effectiveness and safety of modified XYS combined with chemotherapy in-depth, thus providing clinicians and policymakers with evidence-based guidance and new treatment options.
Objective: To comprehensively evaluate the efficacy and safety of modified XYS in conjunction with chemotherapy in treating breast cancer by conducting a meta-analysis.
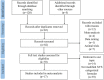
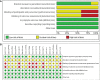
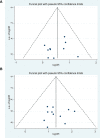
Methods: 8 databases were systemically searched until April 3, 2022, including Web of Science PubMed, EMBASE, Cochrane Library, China National Knowledge Infrastructure (CNKI), Wanfang, Chinese Scientific Journals Database (VIP), and Chinese Biological Medical Database (CBM). Relevant randomized controlled trials (RCTs) comparing modified XYS in combination with chemotherapy versus chemotherapy alone were included. For the evaluation of methodological quality, Cochrane Collaboration was considered. Software Review Manager (version 5.4) was used for data analysis. Software STATA (version 15.0) was employed for sensitivity analysis and publication bias.
Results: Altogether, 17 RCTs involving 1207 patients were investigated in the current review. The findings revealed that modified XYS combined with chemotherapy could lead to beneficial improvements compared to chemotherapy alone. More specifically, the combined therapy could enhance the short-term efficacy in the treatment of solid tumors (OR: 1.74; 95% CI 1.27 to 2.39; P = 0.0006; I2 = 0%); improve QOL (quality of life) (OR: 3.75; 95% CI 2.58 to 5.44; P < 0.00001; I2 = 0%); reduce clinical symptoms (OR: 3.69; 95% CI 1.43 to 9.49; P = 0.007; I2 = 53%); ease depression (MD: -12.96; 95% CI -16.09 to -9.83; P < 0.00001; I2 = 0%); increase leukocytes (OR: 0.32; 95% CI 0.20 to 0.50; P < 0.00001; I2 = 0%) and platelets (OR: 0.37; 95% CI 0.20 to 0.67; P = 0.001; I2 = 0%); reduce nausea and vomiting (OR: 0.26; 95% CI 0.15 to 0.44; P < 0. 00001; I2 = 0%); mitigate cardiotoxicity (OR: 0.16; 95% CI 0.07 to 0.36; P<0.00001; I2 = 0%); prolong survival time (OR: 2.19; 95% CI 1.03 to 4.66; P = 0.04; I2 = 0%), compared to chemotherapy alone. Unfortunately, there was no statistically significant difference in damage to the liver and kidney (OR: 0.59; 95% CI 0.29 to 1.21; P = 0.15; I2 = 0%).
Conclusion: The existing evidence suggests modified XYS combined with chemotherapy leads to beneficial improvements in the management of breast cancer, which may serve as a promising therapy for breast cancer in clinical practice. Given the limited number of high quality RCTs, more rigorous, scientific, double-blinded, large-scale, multi-center clinical trials are warranted further.
Systematic review registration: https://www.crd.york.ac.uk/PROSPERO/, identifier CRD42022357860.
Keywords: Chinese herbal medicine; breast cancer; chemotherapy; meta - analysis; xiaoyao san.
Copyright © 2023 Pan, Fu, Zhou, Lin and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







Similar articles
-
Efficacy and safety of Xiaoyao San in the treatment of chronic fatigue syndrome: a systematic review and meta-analysis.Front Pharmacol. 2025 Feb 6;16:1496774. doi: 10.3389/fphar.2025.1496774. eCollection 2025. Front Pharmacol. 2025. PMID: 39981187 Free PMC article.
-
Safety and efficacy of Xiaoyao-san for the treatment of functional dyspepsia: a systematic review and meta-analysis of randomized controlled trials.Front Pharmacol. 2023 Apr 12;14:1114222. doi: 10.3389/fphar.2023.1114222. eCollection 2023. Front Pharmacol. 2023. PMID: 37124216 Free PMC article.
-
Clinical efficacy and safety of traditional Chinese medicine Xiao Yao San in insomnia combined with anxiety.Medicine (Baltimore). 2021 Oct 29;100(43):e27608. doi: 10.1097/MD.0000000000027608. Medicine (Baltimore). 2021. PMID: 34713840 Free PMC article.
-
Efficacy and Safety of Chinese Herbal Medicine Xiao Yao San in Functional Gastrointestinal Disorders: A meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials.Front Pharmacol. 2022 Jan 20;12:821802. doi: 10.3389/fphar.2021.821802. eCollection 2021. Front Pharmacol. 2022. PMID: 35126152 Free PMC article. Review.
-
External therapy of traditional Chinese medicine for treating irritable bowel syndrome with diarrhea: A systematic review and meta-analysis.Front Med (Lausanne). 2022 Aug 9;9:940328. doi: 10.3389/fmed.2022.940328. eCollection 2022. Front Med (Lausanne). 2022. PMID: 36017012 Free PMC article.
Cited by
-
Mechanism Study of Xiaoyao San against Nonalcoholic Steatohepatitis-Related Liver Fibrosis Based on a Combined Strategy of Transcriptome Analysis and Network Pharmacology.Pharmaceuticals (Basel). 2024 Aug 27;17(9):1128. doi: 10.3390/ph17091128. Pharmaceuticals (Basel). 2024. PMID: 39338294 Free PMC article.
-
Traditional Chinese medicine combined with chemotherapy for breast cancer after operation: A systematic review and meta-analysis.Medicine (Baltimore). 2024 Nov 15;103(46):e40264. doi: 10.1097/MD.0000000000040264. Medicine (Baltimore). 2024. PMID: 39560516 Free PMC article.
-
Unveiling the Mechanisms and Therapeutic Effects of Xiaoyao Sanjie Decoction in Triple-Negative Breast Cancer: A Network Pharmacology and Experimental Validation Approach.Drug Des Devel Ther. 2024 Dec 27;18:6263-6281. doi: 10.2147/DDDT.S492047. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 39741917 Free PMC article.
-
Traditional Chinese medicine in the treatment of breast Cancer.Mol Cancer. 2025 Aug 1;24(1):209. doi: 10.1186/s12943-025-02416-5. Mol Cancer. 2025. PMID: 40750898 Free PMC article. Review.
-
Senkyunolide H reverses depression-induced breast cancer progression by regulating CXCR2.Nat Prod Bioprospect. 2025 Aug 21;15(1):57. doi: 10.1007/s13659-025-00543-6. Nat Prod Bioprospect. 2025. PMID: 40839237 Free PMC article.
References
-
- Chai A, Gan GQ, Xiao L. Analysis of zhang dezhong's skillful use of xiaoyao powder in treating breast cancer. J Hubei Univ Tradit Chin Med (2021) 23(02):112–4.
-
- Zhu XY, Zhang CH, Jia YJ. Professor Wang keqiong's experience in treating breast cancer after radiotherapy with xiaoyao powder. J Tianjin Univ Tradit Chin Med (2021) 40(03):298–300.
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

