Cotton leafroll dwarf disease: An enigmatic viral disease in cotton
- PMID: 37038256
- PMCID: PMC10189767
- DOI: 10.1111/mpp.13335
Cotton leafroll dwarf disease: An enigmatic viral disease in cotton
Abstract
Taxonomy: Cotton leafroll dwarf virus (CLRDV) is a member of the genus Polerovirus, family Solemoviridae. Geographical Distribution: CLRDV is present in most cotton-producing regions worldwide, prominently in North and South America.
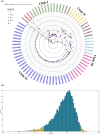
Physical properties: The virion is a nonenveloped icosahedron with T = 3 icosahedral lattice symmetry that has a diameter of 26-34 nm and comprises 180 molecules of the capsid protein. The CsCl buoyant density of the virion is 1.39-1.42 g/cm3 and S20w is 115-127S. Genome: CLRDV shares genomic features with other poleroviruses; its genome consists of monopartite, single-stranded, positive-sense RNA, is approximately 5.7-5.8 kb in length, and is composed of seven open reading frames (ORFs) with an intergenic region between ORF2 and ORF3a.
Transmission: CLRDV is transmitted efficiently by the cotton aphid (Aphis gossypii Glover) in a circulative and nonpropagative manner. Host: CLRDV has a limited host range. Cotton is the primary host, and it has also been detected in different weeds in and around commercial cotton fields in Georgia, USA.
Symptoms: Cotton plants infected early in the growth stage exhibit reddening or bronzing of foliage, maroon stems and petioles, and drooping. Plants infected in later growth stages exhibit intense green foliage with leaf rugosity, moderate to severe stunting, shortened internodes, and increased boll shedding/abortion, resulting in poor boll retention. These symptoms are variable and are probably influenced by the time of infection, plant growth stage, varieties, soil health, and geographical location. CLRDV is also often detected in symptomless plants.
Control: Vector management with the application of chemical insecticides is ineffective. Some host plant varieties grown in South America are resistant, but all varieties grown in the United States are susceptible. Integrated disease management strategies, including weed management and removal of volunteer stalks, could reduce the abundance of virus inoculum in the field.
Keywords: Gossypium hirsutum; Polerovirus; cotton blue disease; cotton leafroll dwarf disease; cotton leafroll dwarf virus.
© 2023 The Authors. Molecular Plant Pathology published by British Society for Plant Pathology and John Wiley & Sons Ltd.
Conflict of interest statement
The authors read the manuscript and declare no conflict of interest.
Figures


References
-
- Aboughanem‐Sabanadzovic, N. , Aboughanem‐Sabanadzovic, N. , Allen, T.W. , Wilkerson, T.H. , Conner, K.N. , Sikora, E.J. et al. (2019) First report of cotton leafroll dwarf virus in upland cotton (Gossypium hirsutum) in Mississippi. Plant Disease, 103, 1798.
-
- Agrofoglio, Y.C. , Delfosse, V.C. , Casse, M.F. , Hopp, H.E. , Kresic, I.B. & Distéfano, A.J. (2017) Identification of a new cotton disease caused by an atypical cotton leafroll dwarf virus in Argentina. Phytopathology, 107, 369–376. - PubMed
-
- Al Rwahnih, M. , Alabi, O.J. , Hwang, M.S. , Tian, T. , Mollov, D. & Golino, D. (2021) Characterization of a new nepovirus infecting grapevine. Plant Disease, 105, 1432–1439. - PubMed
-
- Alabi, O.J. , Isakeit, T. , Vaughn, R. , Stelly, D. , Conner, K.N. , Gaytán, B.C. et al. (2020) First report of cotton leafroll dwarf virus infecting upland cotton (Gossypium hirsutum) in Texas. Plant Disease, 104, 998.
-
- Ali, A. & Mokhtari, S. (2020) First report of cotton leafroll dwarf virus infecting cotton (Gossypium hirsutum) in Kansas. Plant Disease, 104, 1880.
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources

