Corticotropin-releasing hormone neurons in the central nucleus of amygdala are required for chronic stress-induced hypertension
- PMID: 37041718
- PMCID: PMC10325697
- DOI: 10.1093/cvr/cvad056
Corticotropin-releasing hormone neurons in the central nucleus of amygdala are required for chronic stress-induced hypertension
Abstract
Aims: Chronic stress is a well-known risk factor for the development of hypertension. However, the underlying mechanisms remain unclear. Corticotropin-releasing hormone (CRH) neurons in the central nucleus of the amygdala (CeA) are involved in the autonomic responses to chronic stress. Here, we determined the role of CeA-CRH neurons in chronic stress-induced hypertension.
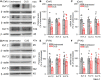
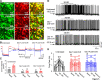
Methods and results: Borderline hypertensive rats (BHRs) and Wistar-Kyoto (WKY) rats were subjected to chronic unpredictable stress (CUS). Firing activity and M-currents of CeA-CRH neurons were assessed, and a CRH-Cre-directed chemogenetic approach was used to suppress CeA-CRH neurons. CUS induced a sustained elevation of arterial blood pressure (ABP) and heart rate (HR) in BHRs, while in WKY rats, CUS-induced increases in ABP and HR quickly returned to baseline levels after CUS ended. CeA-CRH neurons displayed significantly higher firing activities in CUS-treated BHRs than unstressed BHRs. Selectively suppressing CeA-CRH neurons by chemogenetic approach attenuated CUS-induced hypertension and decreased elevated sympathetic outflow in CUS-treated BHRs. Also, CUS significantly decreased protein and mRNA levels of Kv7.2 and Kv7.3 channels in the CeA of BHRs. M-currents in CeA-CRH neurons were significantly decreased in CUS-treated BHRs compared with unstressed BHRs. Blocking Kv7 channel with its blocker XE-991 increased the excitability of CeA-CRH neurons in unstressed BHRs but not in CUS-treated BHRs. Microinjection of XE-991 into the CeA increased sympathetic outflow and ABP in unstressed BHRs but not in CUS-treated BHRs.
Conclusions: CeA-CRH neurons are required for chronic stress-induced sustained hypertension. The hyperactivity of CeA-CRH neurons may be due to impaired Kv7 channel activity, which represents a new mechanism involved in chronic stress-induced hypertension.
Keywords: Central nucleus of the amygdala; Chronic stress; Corticotropin-releasing hormone; Hypertension; Kv7 channel.
© The Author(s) 2023. Published by Oxford University Press on behalf of the European Society of Cardiology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
Conflict of interest: none declared.
Figures





Similar articles
-
Impaired Kv7 channel activity in the central amygdala contributes to elevated sympathetic outflow in hypertension.Cardiovasc Res. 2022 Jan 29;118(2):585-596. doi: 10.1093/cvr/cvab031. Cardiovasc Res. 2022. PMID: 33512443 Free PMC article.
-
Signaling pathways involved in NMDA-induced suppression of M-channels in corticotropin-releasing hormone neurons in central amygdala.J Neurochem. 2022 Jun;161(6):478-491. doi: 10.1111/jnc.15647. Epub 2022 May 30. J Neurochem. 2022. PMID: 35583089 Free PMC article.
-
Hypothalamic Corticotropin-Releasing Hormone Contributes to Hypertension in Spontaneously Hypertensive Rats.J Neurosci. 2023 Jun 14;43(24):4513-4524. doi: 10.1523/JNEUROSCI.2343-22.2023. Epub 2023 May 9. J Neurosci. 2023. PMID: 37160364 Free PMC article.
-
Corticotropin-releasing hormone neurons in the central Amygdala: An integrative hub for pain, emotion, and addiction neurobiology.Brain Res. 2025 Sep 15;1863:149753. doi: 10.1016/j.brainres.2025.149753. Epub 2025 Jun 2. Brain Res. 2025. PMID: 40467025 Review.
-
Corticotrophin-releasing factor 1 activation in the central amygdale and visceral hyperalgesia.Neurogastroenterol Motil. 2015 Jan;27(1):1-6. doi: 10.1111/nmo.12495. Neurogastroenterol Motil. 2015. PMID: 25557223 Free PMC article. Review.
Cited by
-
Relations between neurometabolism and clinical biomarkers in patients with metabolic disease.Front Neurosci. 2025 Apr 15;19:1547010. doi: 10.3389/fnins.2025.1547010. eCollection 2025. Front Neurosci. 2025. PMID: 40303609 Free PMC article.
-
Cardiac vagal motor neurons.Curr Opin Neurobiol. 2025 Aug;93:103068. doi: 10.1016/j.conb.2025.103068. Epub 2025 Jun 25. Curr Opin Neurobiol. 2025. PMID: 40570503 Review.
-
Relationship Between Occupational Noise Exposure and Gestational Hypertension in Pregnant Women in Taizhou City: Cross-Sectional Study.Noise Health. 2024 Apr-Jun 01;26(121):214-219. doi: 10.4103/nah.nah_62_24. Epub 2024 Jun 21. Noise Health. 2024. PMID: 38904825 Free PMC article.
-
Comprehensive Brain-wide Mapping of Afferent and Efferent Nuclei Associated with the Heart in the Mouse.Neurosci Bull. 2025 Mar 15. doi: 10.1007/s12264-025-01384-6. Online ahead of print. Neurosci Bull. 2025. PMID: 40089653
-
Association of weekend warriors and other physical activity patterns with hypertension in NHANES 2007-2018.Sci Rep. 2025 Mar 24;15(1):10042. doi: 10.1038/s41598-025-95402-2. Sci Rep. 2025. PMID: 40122925 Free PMC article.
References
-
- Singh M, Singh AK, Pandey P, Chandra S, Singh KA, Gambhir IS. Molecular genetics of essential hypertension. Clin Exp Hypertens 2016;38:268–277. - PubMed
-
- Friedman R, Iwai J. Genetic predisposition and stress-induced hypertension. Science 1976;193:161–162. - PubMed
-
- Matthews KA, Katholi CR, McCreath H, Whooley MA, Williams DR, Zhu S, Markovitz JH. Blood pressure reactivity to psychological stress predicts hypertension in the CARDIA study. Circulation 2004;110:74–78. - PubMed
-
- Kanayama N, Tsujimura R, She L, Maehara K, Terao T. Cold-induced stress stimulates the sympathetic nervous system, causing hypertension and proteinuria in rats. J Hypertens 1997;15:383–389. - PubMed
-
- Grotto I, Grossman E, Huerta M, Sharabi Y. Prevalence of prehypertension and associated cardiovascular risk profiles among young Israeli adults. Hypertension 2006;48:254–259. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

